1. I. Introduction
mong dental emergencies that the odontosto matologist has to face in his daily practice, circumscribed cellulitis of dental origin play an important role because of the pain they cause and especially the speed with which they can appear and develop. 1 It is an infection of the cellular spaces of the face whose origin is a dental, peri-dental organ. 2,3 Decreasing since the advent of antibiotics, 1 they are still common in developing countries such as Cameroon with a prevalence of 7.4% . 2,4 Cellulitis can be divided into two groups: diffuse cellulitis often serious, sometimes life-threatening and cellulitis circumscribed, less formidable. Their medication management is poorly codified in our context because it is based on the sole bacteriological presumption. 5 The consequence that follows is the advent of bacterial resistance. l'association amoxicilline-métronidazole est largement utilisé dans le traitement des infections odontogènes. Par conséquent, cette étude était conçue pour générer des données à l'appui de son utilisation. Il s'agissait de démontrer l'efficacité clinique de l'association amoxicilline-métronidazole par rapport à l'association spiramycine-métronidazole chez les sujets ayant une cellulite circonscrite odontogène. L'essai clinique était complété par une évaluation de l'observance et de la tolérance des antibiotiques à l'étude. Méthodologie : Il s'agissait d'un essai clinique, randomisée, parallèle, en double insu. Cette étude évaluait l'efficacité, la tolérance et l'observance des associations amoxicillinemétronidazole et spiramycine-métronidazole chez des sujets adultes victimes d'une cellulite circonscrites odontogènes. Les sujets étaient réévalués après trois, cinq et sept jours. A la fin du traitement, les patient's ont été classés en (succès clinique) ou « échec ».
Résultats : L'étude a été menée sur 81 patients en analyse en intention de traiter et 61 patients en analyse per-protocole. En intention de traiter, le succès clinique des associations amoxicilline-métronidazole, spiramycine-métronidazole était supérieure à 90 % dans le traitement des cellulites circonscrites séreuses. La tolérance clinique et l'observance n'étaient pas différentes d'un groupe à un autre.
Conclusion: La non-inferiority de l'association amoxicillinemétronidazole par rapport à l'association spiramycinemétronidazole a été démontrée en analyse en intention de traiter et en per-protocole.
Mots clés: Cellulite, amoxicilline, spiramycine, métronidazole, per-protocole, intention de traiter.
2. Thus, the French Agency for Sanitary Safety of Health Products (AFSSAPS) recommends first-line
In the current, we tested the hypothesis of the non-inferiority of the amoxicillin / metronidazole combination compared to the spiramycin / metronidazole combination in the treatment of odontogenic circumscribed cellulitis. The objective of this study was therefore to evaluate in a clinical study, the efficacy, safety and compliance of the oral amoxicillin-metronidazole combination and of another reference association, namely the spiramycin / metronidazole combination.
3. II. Methodology a) Description of the clinical trial
This was a multicenter, double-blind, randomized, non-inferiority clinical trial in two patient groups. The study was carried out for five months, from January 9, 2017 to May 29, 2017, in Cameroon in odontostomatology departments of: University Teaching and Hospital Center of Yaounde, Central Hospital of Yaounde, Military Hospital of Yaounde and the Jamot Hospital of Yaounde. The study was conducted according to the guidelines on good clinical practices, the Declaration of Helsinki (2008). The risk analysis was carried out as part of the protocol development. The study protocol has been approved by the Ethics Committee of the Faculty of Medicine and Biomedical Sciences (FMSB).The trial included a principal investigator and four secondary investigators from the previously mentioned hospitals.
4. b) Eligibility criteria
Any patient over the age of 18 who had given informed consent was likely to be included as soon as he had evidence of odontogenic circumscribed cellulitis (pain, swelling) with or without suppuration, trismus, or 'enanthema. The non-inclusion criteria were: cellulitis fistulized to the skin, diffuse or diffused perimaxillary cellulitis, ? A severe pathology that may not allow an evaluation of the efficacy and the tolerance of the treatment under study, ? Pregnant or lactating women, ? Patients with contraindications specific to the studied drugs ? Anyone who has received antibiotic therapy in the previous two weeks. ? Patients treated with an anti-inflammatory
5. c) Assessment of infection / inflammation
Clinical data were collected at baseline, at reassessment visits, and at the end of treatment. They included general (fever, asthenia) and locoregional signs of inflammation and infection: pain, edema, suppuration, trismus, lymphadenopathy, enanthema.
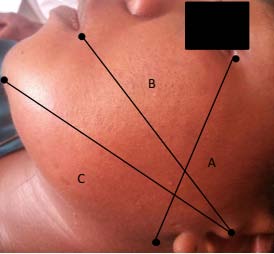
The trismus was quantified according to a scale of gravity comprising 4 classes: absent (mouth opening 3 fingers), medium (2 fingers), large (1 finger crossing), and total (0 finger crossing). The size of the edema was measured using the tape measure method [10]. Three measurements were obtained by five reference points ipsilateral to the lesion: the tragus, the cutaneous pogonion, the external can thus of the eye, the gonion, the labial commissure (figure1).The sum of the three measurements was the average of the edema size for that day.
The pain was graduated by the simple verbal scale. It is an ordinal scale, usually consisting of five ordered categories of verbal descriptors. Each is related to a score from 0 to 4. Absence of pain = 0; Slight pain = 1; Moderate pain = 2; Severe pain = 3; Unsupportable pain = 4.
The presence or absence of an enanthema was also noted.
6. d) Outcome measures
At the end of the treatment, the patients were classified by the investigator as "cure" (clinical success) or "failure" according to precise rules.
The primary outcome retained was the success rate of treatment. Success was defined by the disappearance of edema and pain after seven days of antibiotic therapy, the absence of suppuration (suppurative cellulitis), trismus, enanthema with respect to inclusion. monotherapy and second-line combination antibiotics according to the severity of infection. 6,7 Among other recommended combinations, amoxicillin/metronidazole combination has bacteriological and pharmacokinetic characteristics to consider its use in circumscribed cellulitis of dental origin. 8,9 Also in a precarious economic and social environment, the use of this combination should reduce the cost of taking care of odontogenic cellulitis because it is accessible and inexpensive.
The secondary outcomes were on the one hand a reduction of at least 70% of the pain; a reduction of edema; a trismus score decreased by at least two levels; the absence of adverse effects.
7. e) Sample size
As for an equivalence study, the number of subjects to be included was estimated at 138 evaluable patients per group, based on the following assumptions: 90% expected success rate, 10% equivalence threshold, risk 5% ? and 20% risk of ? in unilateral formation. This corresponded to a total of 276 patients to include.
8. f) Randomization and blinding
The patient was taken to the investigator of each center, who was responsible for the random assignment of drugs. The patient should not inquire about the treatment to be taken, nor to do anything to know the same for the secondary investigators. Each patient randomly drew two vials in non-transparent bags. These contained 10 to 12 vials per center. Once the draw was done, the bottle was no longer put in the bag (draw without discount) and the patient was put under the treatment carried by the draw. The prints were made successively by the selected patients until the last bottle. Beforehand, the drugs were removed from their packaging to be inserted in bottles of different colors. Green for amoxicillin or spiramycin and red for metronidaz ole. Each patient included had to be treated with an antibiotic combination for seven days and received:
either the spiramycin / metronidazole combination (3 MIU / 500mg): one tablet, twice a day for 7 days), or the amoxicillin / metronidazole combination (1000mg / 500mg): one amoxicillin tablets, twice daily and one tablet of metronidazole twice daily for7 days). GroupI corresponded to the amoxicillin / metronidazole combination and group II to the spiramycin / metronidazole combination.
Standard local treatment was performed at 72 hours. It consisted in carrying out either an extraction, associated or not with a drainage or a canal treatment, associated or not with a drainage.
Finally, each subject had to undertake to perform another 03 visits planned for the test: day3; day5; day7.These control visits made it possible to judge the disappearance or the preservation of the signs present at the time of inclusion, the presence or absence of signs of drug intolerance and finally the existence or not of a relapse. Tolerance and compliance were assessed at the level of both groups.
9. g) Statistical tests
The quantitative variables were compared by a Student's test, and the qualitative variables by a Chi 2 test. The comparison of the efficacy of the treatments was carried out using a test of equivalence in unilateral formation. The non-inferiority of spiramycin / metronidazole was considered to be demonstrated if the confidence interval was included within [-? ; 10%], the equivalence threshold being set at 10%.The analysis was performed using ? risk threshold of 5%. The analysis was performed in the treatment intent (ITT) population and the per-protocol population (PP).
10. III. Results
11. a) Populations and treatments
We selected 86 patients who were randomized. Five were excluded because they were lost after the inclusion visit. The remaining 81 cases were analyzed for intent to treat (41 in group I and 40 in group II). The per-protocol analysis led us to exclude 20 patients for non-compliance. Thus, the study was conducted on 61 patients (32 in group I and 29 in group II) in per-protocol analysis.
12. Figure 2: Chronology of the clinical trial b) Baseline characteristics
The population included 47 men and 37 women with a mean age of 34 ± 14 years and an average weight of 72.2 ± 9.2 kg with no difference between the two groups (ITT). Regarding the signs and symptoms at baseline, no significant differences were noted between the two groups. Pain, enanthema and edema were present in all cases. Suppuration in 41 cases, lymphadenopathy in 64 cases, trismus in 40 cases, most often of mild or significant intensity. The general characteristics are presented in Table I.
13. ITT: Intent to treat c) Primary outcomes ? Serous Cellulitis
The percentage of patients do not show clinical signs from the third visit was comparable in both groups (100% and 95% for group I and II respectively in intent to treat, 100% in the per-protocol for group I and II). Table II illustrates the success rate and failures after taking antibiotics. The difference in treatment between the two groups was 5% in intention of treatment with confidence interval [-3.8%, 9.4%].Table II illustrates clinical success and failure rates after taking antibiotics.
14. Table II: Clinical
? Suppurative Cellulitis Clinical success from intent-to-treat analysis was 85% in group I and 80% in group II. Table III details the clinical success and failure rates after taking antibiotics.
Table III: Success and Failures in the Treatment of Suppurative Cellulitis (Day 7)
ITT: intent to treat; PP: per-protocol d) Secondary outcomes ? Pain: The average pain was reduced more than 70% from one group to the other inclusion at the end of treatment. This reduction is illustrated in Figure 3 below.
15. ? Edema:
The mean edema at baseline was 18.5 ± 1.7 cm in group I versus 18.4 ± 1.5 cm in group II (P = 0.66) The average of the edema on the seventh day was 12.9 ± 1 and 12.4 cm ± 3.6 cm from one group to another. P = 0.15 ? Trismus: On the seventh day trismus three finger was present in 41 patients in group I and 36 patients in group II.
16. e) Clinical tolerance of antibiotics
A total of 47 side effects were reported, 20 in group I and 27 in group II. The most frequently observed adverse reactions with a frequency ?3% wer e nausea, candidacies, epigastralgia and headache. The incidence of side effects was similar between treatment groups, with the exception of nausea that was reported in 37% of patients in group II.
17. f) The observance
Treatment compliance was consistent in 32 patients, or 78% of Group I (p= 0.396). Treatment compliance was non-compliant in 30 patients, 73.2% of group II. P = 0.811
18. IV. Discussion
In order to overcome the subjectivity of the Verbal Rating Scale (VRS), only subjects who showed a 70% decrease in pain were considered in calculating the response to clinical success. The required number of patients was not reached due to a strict methodology, refusal and / or delay of some hospital structures to allow this study and finally the time allowed for it was reduced. The percentage lost to follow-up did not exceed 10%. Proportion beyond which the interpretation of the results becomes prejudicial.
The primary objective was to evaluate the clinical efficacy of two antibiotic treatments of which only one, the spiramycin-metronidazole combination, had been the subject of previous studies already cited in the literature .This led to the choice of a statistical analysis called "non-inferiority", the spiramycin-metronidazole combination being used as a reference. These antibiotics are the most prescribed by Cameroonian dental doctors. 11 In serous cellulitis, the clinical success rate was higher than the expected 90% per-protocol and intention-to-treat ratio in both groups. The non-inferiority amoxicillin / metronidazole combination is demonstrated because the difference between the two antibiotics is less than the allowed threshold (10%) and the confidence interval is included in the limits [-?; 10%]. This result is similar to that of Mello et al who achieved a clinical success rate of 85% for amoxicillin and 89.2% for the spiramycin / metronidazole combination in the treatment of pericoronitis.12 This is explained by the fact that aerobic microbial flora predominates in circumscribed cellulite .5This flora would be highly sensitive to amoxicillin. Amoxicillin is a bactericidal antibiotic whose broad spectrum of activity includes most aerobic and anaerobic bacteria in the oral cavity . 8 However, its important use for years led to the selection of natural resistant or related to the production of betalactamases, questioning its use as monotherapy. 13,14 Metronidazole, derivative of 5-nitroimidazole, is a bactericidal agent.15 Its spectrum of activity is carried on most anaerobic strains associated with oral infections, including those found in abscesses of dental origin.16 This activity is particularly useful for producing anaerobic bacteria bêta-lactamase. 13,[15][16][17] The combination of amoxicillin and metronidazole, because of the complementarily of the spectra of antibacterial activity, also makes it possible to cover most of the strains present in serous cellulites. Additionally, simultaneous administration of both antibiotics can be accompanied by a synergistic effect. In suppurative cellulitis, success rates were lower than expected in both intention-to-treat groups. This result is the consequence of the inclusion of patients with both non-compliant compliance and high failure rate.
In addition, inadequate and inappropriate ant biotherapy significantly modifies the bacteriology of perimaxillary cellulitis. 18 Although it is a protective factor, inappropriate antibiotic therapy may be a factor in the progression of infection because it is administered at sub-therapeutic doses. Inappropriate and abusive antibiotic therapy promotes the proliferation of antibioticresistant pathogenic bacteria. 18 Pseudomonas aeruginosa, enterobacteria and Acinetobacter baumannii, often found in odontogenic cellulitis are known for their high resistance to certain antibiotics (spiramycin, metronidazole, amoxicillin, amoxicillin/ clavulanic acid). 13,[18][19][20] Their resistance may be due to the existence of a selection pressure exerted by the antibiotics. Nevertheless, the confidence interval is included in the bounds [-?; 10%]. Thus, we can conclude that amoxicillin / metronidazole is non-inferior.
A higher percentage of the pain reduction (95.1%) was achieved in group I compared to group II (84%) after five days of treatment. In a similar study to demonstrate possible differences in the severity of symptoms after using amoxicillin and spiramycin/metronidazole in péricoronarites, pain was considered less acute with amoxicillin.12Thus, these results indicate that the amoxicillin / metronidazole combination administered twice daily for the treatment of circumscribed cellulitis serves as an appropriate treatment option with the potential benefit of an early clinical response.
19. V. Conclusion
The results of our study confirm the equivalence of efficacy, tolerance and compliance of the amoxicillin/ metronidazole combination compared to the spiramycin/ metronidazole combination. A seven-day treatment with amoxicillin/metronidazole (1000mg / 500mg) with four tablets for amoxicillin and two tablets for metronidazole a day is therefore as effective in the treatment of odontogenic circumscribed cellulitis as the combination spiramycin/metronidazole. Conflicts of interest: None
| General characteristics | Group I | Group II | p | ||||
| Mean age +/-SD (years) | 34± 15 | 33 ± 14 | 0.68 | ||||
| Sex | |||||||
| Male | 25 | 22 | |||||
| Female | 16 | 18 | 0.41 | ||||
| Average weight +/-SD (Kg) | 72.7 ± 9,7 | 71.8 ± 9.9 | 0.84 | ||||
| Average size +/-SD (cm) | 169 ± 9 | 169 ± 10 | 0.68 | ||||
| Signs and symptoms | n | % | n | % | n | % | |
| pain | 41 | 100 | 40 | 100 | 81 | 100 | 0.25 |
| edema | 41 | 100 | 40 | 100 | 81 | 100 | 0.34 |
| Trismus | |||||||
| Absent | 20 | 48.8 | 17 | 42.5 | 37 | 45.7 | |
| Mild | 11 | 26.8 | 12 | 30 | 23 | 2.3 | |
| Important | 09 | 22 | 08 | 20 | 17 | 21 | |
| Total | 01 | 2.4 | 03 | 7.5 | 04 | 05 | |
| Lymphademopathy | |||||||
| 33 | 80.4 | 31 | 77.5 | 64 | 79 | ||
| Suppuration | 23 | 56 | 18 | 45 | 41 | 50.6 | 0.15 |
| Enanthema | 41 | 100 | 40 | 100 | 81 | 100 | |
| Clinical forms | |||||||
| Cellulitis | |||||||
| Serous | 21 | 20 | 41 | ||||
| suppurative | 20 | 20 | 40 | ||||