1. Introduction
iabetes mellitus is a pandemic disease that has exponentially increasing prevalence. In 1980, 108-million persons had diabetes worldwide while in 2014, 430-million persons were affected [1]. In spite of the increased awareness about this disease and the worldwide efforts to give optimum care, 3.7 million diabetic patients lost their lives in 2012 because of diabetes and its complication [2]. This figure exceeded 5 million deaths annually in the last few years [3]. The hazard of cardiovascular mortality among diabetic patients is 2.3 folds that in non-diabetic personnel [4]. The average life span of the diabetic patients is 10-15-years shorter than non-diabetic subjects [5]. Besides, diabetes is the cause of many disabling Besides, diabetes is the cause of many disabling morbidities. In spite of the optimal management of the established cases of diabetic retinopathy that reduces the risk of visual loss by 60%, diabetes remains D the leading cause of blindness among working-age adults worldwide [6]. Diabetes is the leading cause of non-traumatic lower-extremity amputation [7]. Diabetic peripheral neuropathy (PN) is the most frequent cause of sensory neuropathy [8]. Diabetic kidney disease (DKD) is the most common cause of end-stage renal disease (ESRD). One-third of T1DM develop ESRD, while only 10-20% of type 2 diabetes mellitus (T2DM) patients progress to ESRD [9,10]. The prevalence of congestive heart failure (CHF) among diabetic patients aged 55 to 64 years is 5.5 folds the prevalence among nondiabetic personnel of the same age [11]. Diabetes is an independent risk factor for the development of ischemic heart disease (IHD). CHF and IHD are the commonest causes of death in T1DM and T2DM patients [12]. Diabetes mellitus confers a high risk of cerebrovascular stroke [13]. Endothelial dysfunction is a common pathology underlying the etiopathogenic mechanism of all these complications [14]. This endothelial dysfunction is a sequel to many metabolic changes encountered in hyperglycemic personnel. These metabolic changes include increased oxidative stress [15], hyperuricemia [16], stimulation of sodium hydrogen exchangers (NHE) [14], and stimulation of renal sodium glucose transporters (SGLT) [17].
Twenty-five years ago, the Diabetes Control and Complications Trial (DCCT) research group announced 50% reduction of microvascular complications among T1DM patients in the tight blood sugar control group compared to poorly controlled cases [18]. Five years later, the United Kingdom Prospective Diabetes Study (UKPDS) group announced similar findings among T2DM patients [19]. However, tight blood sugar control only had a marginal impact on cardiovascular disease and all-cause mortality among diabetic patients [20]. Additionally, blood sugar control using sulphonylurea compounds and insulin carries increased risk of severe hypoglycemia and weight gain [18,19]. IN UKPDS study, T2DM patients allocated to metformin had 32% reduction for any diabetes-related endpoint, 42% for diabetes-related death, and 36% for all-cause mortality when compared with those prescribed sulphonylurea or insulin [21]. These favorable effects of metformin were attributed to body weight reduction and the almost absence of hypoglycemic attacks. According to these results and others, the American College of Endocrinology (ACE), and the American Association of Clinical Endocrinology (AACE) recommend that the choice of anti-diabetic therapies must depend on many attributes that include antihyperglycemic efficacy; risk of inducing hypoglycemia; and risk of weight gain [22]. The last 15 years have witnessed the introduction of three new hypoglycemic agents, namely, glucagon-like peptide-1 receptor agonists (GLP-1RA), dipeptidyl peptidase 4 inhibitors (DPP4Is), and sodium glucose cotransporter-2 inhibitors (SGLT2Is). These three agents carry unique features, namely, the minimal incidence of hypoglycemic events and the favorable impact on body weight. GLP-1RA and SGLT2Is are associated with body weight reduction, while DPP4Is are weight neutral [23,24]. Compared to older hypoglycemic agents, these newer groups carry potential favorable protective effects on endothelium, and can significantly reduce adverse cardiovascular events of diabetes in case of SGLT2Is and GLP-1RA, and are reno-protective. SGLT2Is mayalso prevent or withhold diabetic retinal complications [25]. This review will highlight the possible new strategy to prevent the development and progression of diabetic complications, the main target of this disease management.
2. a) The Endothelium in Diabetes
The first report on the role of the endothelium as an important regulator of local vascular tone was in 1980 [26]. The vascular endothelium is an important component of diabetic complications. Endothelial dysfunction is eminent not only in diabetic patients but also in patients suffering obesity or metabolic syndrome. Decreased synthesis of nitric oxide (NO), a potent vasodilator, is the salient feature of endothelial dysfunction. Decreased NO underlies insulin resistance by reducing insulin access to target cells [27]. Insulin has also to cross endothelial cells to reach target tissues [28,29]. Hyperglycemia can lead to endothelial mitochondrial fragmentation and increased production of reactive oxygen species (ROS) [30]. Increased endothelial ROS is associated with increased breakdown of NO [31]
3. b) Sodium Hydrogen Exchangers
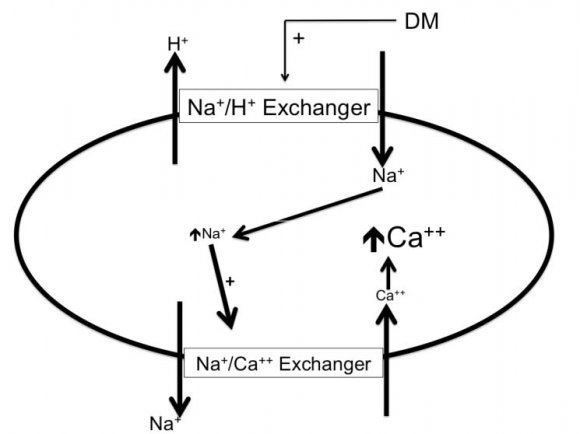
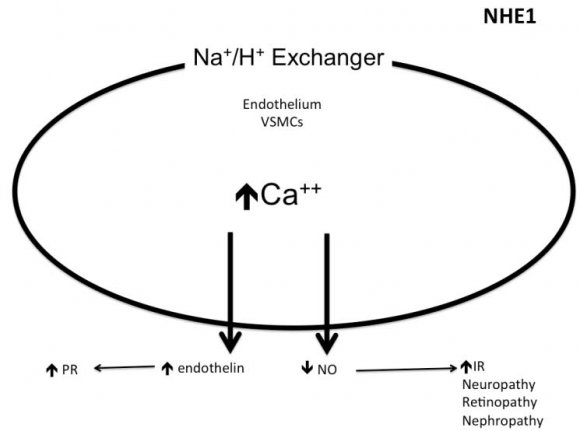
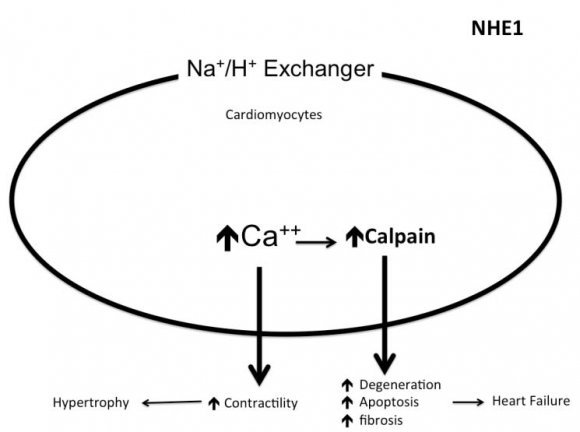
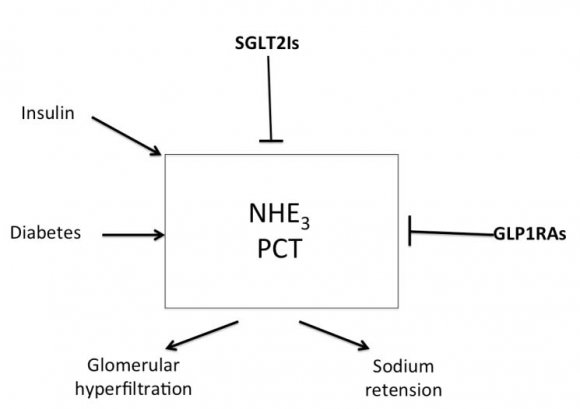
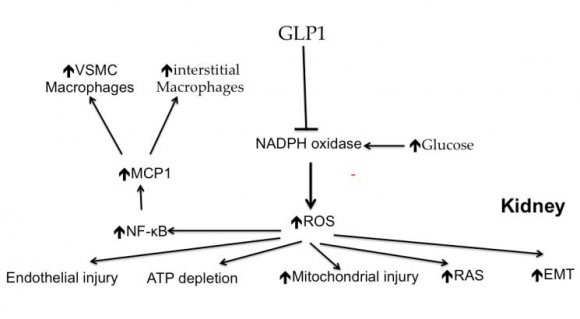
The sodium hydrogen exchangers (NHE) are trans-membrane ion channels that are responsible for intracellular pH regulation through the extrusion of hydrogen in exchange with sodium influx [Fig. 1]. NHE exist in nine isoforms [41,42]. NHE1 is present on the surface of endothelium, vascular smooth muscle cells (VSMCs), cardiomyocytes and platelets, while in case of renal tubular and intestinal epithelium NHE3 is encountered. Activation of the NHE1 within endothelium, VSMCs, and cardiomyocytes may underlie microvascular and macro-vascular complications of diabetes. It can also have a role in insulin resistance and systemic hypertension. These exchangers cause increased sodium influx that stimulates sodium-calcium exchanger with consequent increase of intracellular calcium. Within endothelium, increased cytoplasmic calcium inhibits eNOS and thus decreases NO synthesis (Fig. 2). Increased intracellular calcium isalso associated with increased intracellular and mitochondrial activity of calpain, the cysteine protease, that can damage the inner mitochondrial membrane, a process that ends with cell apoptosis [43]. Activation of NHE1 in diabetic patients is a consequence of high blood glucose, insulin, angiotensin, or adipokines [44]. Endothelial NHE1 activation increasesthe influx of calcium into the cytoplasm and mitochondria associated with increased calpain enzyme activity. These changes lead to endothelial dysfunction and senescence. The development of systemic hypertension, increased insulin resistance, diabetic retinopathy, nephropathy, and neuropathy are consequences of decreased eNOS activity and accelerated endothelial senescence. It can also explain the increased frequency of vascular calcification, peripheral vascular disease, and diabetic cerebrovascular dysfunction [45]. Mitochondrial injury is associated with impaired antioxidant defense [46]. Inhibition of NHE1 using cariporide was associated with increased NO release; eNOS activity simultaneously decreased ROS production, decreased nuclear factor-?B (NF-?B) activation and decreased the production of tumor necrosis factor-? and intercellular adhesion molecule-1 [47]. Increased intracellular calcium induced by NHE1 isoform on the surface of cardiomyocytes leads to cardiac hypertrophy. Peripheral coronary ischemia secondary to endothelial dysfunction can further activate cardiac NHE1.Active NHE1 increases intracellular and intra-mitochondrial calpain that contributes to degeneration, apoptosis, and fibrosis of myocardium [44] [Fig. 3]. Activation of renal NHE3 within PCT and ascending loop of Henle causes sodium retention and can thus contribute to the development of systemic hypertension in diabetic patients [44,48] [Fig. 4]. Activation of NHE1 on the surface of platelets plays a significant role in platelet activation. This effect is mediated through increased intracellular calcium and can contribute to the pro-coagulant state in diabetes [49]. Accordingly, activation of NHE1 on the surface of endothelial cells, VSMCs, platelets, and cardiomyocytes Increased oxidative stress is one of the metabolic disorders encountered in diabetes. Diabetic patients have overproduction of free oxygen radicals and decreased wash out of these radicals. Increased production of free oxygen radicals is the sequel to increased activity of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase [50,51], cyclo-oxygenase [52], and lipoxygenase [53] enzymes. Hyperglycemia stimulatesall these enzymes. Sodium-glucose cotransporter 2 (SGLT2) within the brush border of the proximal convoluted tubular epithelium (PCT) is another pathway of free oxygen radicals' overproduction. Increased intracellular uric acid (UA) induces NADPH oxidase [54]. Mitochondrial damage results in impaired antioxidant defense [46]. Increased free oxygen radicals activate NF-?B [55]. When NF-?B is free from its inhibitor, it translocates from the cytoplasm to the nucleus where it triggers the genes encoding transforming growth factor-?1 (TGF-?1), and monocyte chemo attractant protein-1 (MCP-1) and Intercellular Adhesion Molecule 1 (ICAM1) [56][57][58]. Reactive oxygen species (ROS) stimulate overproduction of protein kinase C (PKC) and mitogen-activated protein (MAP) kinase within mesangial cells (MCs) and pericytes. All these factors stimulate overproduction of extracellular matrix proteins [59].
4. d) Uric acid
Serum uric acid (UA) is a strong predictor for the development of proteinuria in T1DM patients. The risk for proteinuria increases by 80% with every 1mg/dL rise in serum UA [60]. The risk of decline of glomerular filtration rate (GFR) is significantly higher (2.4 folds) in T1DM patients with serum UA>6.6 mg/dL when compared with candidates with lower level [61]. In T1DM patients followed-up for more than 18 years, serum UA was an independent predictor of overt proteinuria [62]. In T2DM patients, 68% of the hyperuricemic versus 41.5% with normal serum UA had diabetic nephropathy (DN) [63]. Further prospective studies confirmed the increased risk of development of proteinuria and accelerated decline of GFR among hyperuricemic T2DM [64,65]. Serum UA> 7mg/dL in males and > 6mg/dL in females were associated with a higher rate of DN progression, and overall mortality among T2DM patients that have the disease for fifteen years or more [66]. Treatment of T2DM patients suffering DN and high serum UA with allopurinol was associated with a significant decrease of urine albumin excretion (UAE) and serum creatinine and a significant increase of GFR over three years of follow-up [67]. A recent metaanalysis of 19 randomized controlled trials has confirmed the significant favorable effect of uratelowering therapy on the rate of GFR decline. These 19 trials enrolled 992 patients [68].
Increased level of Serum UA is associated with endothelial dysfunction. High mobility group box chromosomal protein 1 (HMGB1) is a pro-inflammatory mediator synthesized and secreted by activated phagocytes or monocytes. When secreted extracellular, HMGB1 can interact with the receptor for advanced glycation end products (RAGE), inducing the production of multiple cytokines, and the induction of vascular adhesion molecules [69]. In a recent in vitro study, high UA concentration inhibited eNOS expression and NO production in human umbilical vein endothelial cells (HUVECs), increased extracellular HMGB1 secretion, up-regulated RAGE expression, activated NF-?B, and increased the level of inflammatory cytokines. Blocking RAGE significantly suppressed the DNA binding activity of NF-?B and the levels of inflammatory cytokines [70]. HighserumUA is also a significant predictor of systemic hypertension [71].
5. e) Role of glucagon like peptide-1 receptor agonists (GLP-1RA)
Glucagon-like peptide-1 (GLP-1), is a peptide hormone secreted by the neuro-endocrine cells within the mucosa of the small intestine [72]. In healthy individuals, GLP-1 activates insulin secretion, inhibits glucagon secretion and slows gastric emptying and controls apetite [72]. The susceptibility of this peptide hormone to enzyme breakdown by the dipeptidyl peptidase-4 enzyme (DPP-4) is responsible for the very short plasma half-life of GLP-1. It cannot be used therapeutically except as continuous intravenous infusion [73]. GLP-1RA are exogenous GLP-1 analogues with variable sequence similarity to the human GLP-1 [74]. The variability involved mainly two sites in the GLP-1 molecule susceptible to cleavage by DPP4; namely, alanine and lysine at positions 8 and 34 respectively. These changes, beside other modifycations, have helped to find out many peptides that simulate GLP-1 action but with longer half-life [73]. GLP-1RAs were found to decrease body weight, and some cardiovascular morbidity, without increasing the risk of hypoglycemia [75]. Robust indications for GLP-1RAs in T2DM patients not responding to metformin monotherapy, dual therapy, or insulin include overweight, inability to control appetite, high risk of cardiovascular disease, and the need of high doses of insulin [73]. The use of GLP-1 RAs can also lower systolic, and to a minor degree, diastolic blood pressure [76]. However, long term use of GLP-1 RAs was frequently reported to be associated with increased heart rate [76,77]. The current evidence does not support any beneficial effect of GLP-1RAs in patients with heart failure and/or impaired ventricular function [78,79]. The Evaluation of Lixisenatide in Acute Coronary Syndrome (ELIXA) trial was the first cardiovascular outcome trial (CVOT) of GLP-1RAs in T2DM. Based on this trial, treatment with lixisenatide in addition to conventional therapy had no impact on the cardiovascular risk in patients with T2DM and recent acute coronary syndrome [80]. In the Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial, that appeared in 2016, liraglutide use significantly decreased mortality from any cause and cardiovascular events in patients with T2DM at high risk for cardiovascular events. The benefit of liraglutide treatment is more prounced patients with eGFR <60 mL/min/1.73 m 2 and patients aged 50 years or more. In spite of these benefits, hospitalization rate for heart failure was not different between liraglutide and placebo groups [81]. Although the incidence of
6. f) Dipeptidyl peptidase 4 inhibitors
The discovery of non-enzymatic functions for DPP4 within the kidney has attracted the attention for the reno-protective action of this hypoglycemic agent especially after disclosure of the antiproteinuric effect of saxagliptin in T2DM patients during "Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus -Thrombolysis in Myocardial Infarction 53" (SAVOR-TIMI 53) trial [87][88][89][90][91]. Experimental pharmacologic and genetic inhibition of DPP4 had also proven efficacy in preventing progressive renal damage in animal models of acute and chronic kidney disease [92,93].
The glucose-lowering action of DPP4Is is through inhibition of breakdown of endogenous GLP and glucose-dependent insulinotropic peptide (GIP). These incretins improve the sensitivity of pancreatic ? cells to glucose [94]. DPP4 exists in 2 forms; membrane-bound and soluble forms [95]. Membranebound DPP4 is present on the cell membrane of epithelial cells in the kidneys, lungs, and small intestine. It also exists on endothelial, and immune cells [96][97][98]. DPP4 on the surface of immune cells is also known as cluster of differentiation 26 (CD26) [97,98]. The soluble form (sDPP4) is the consequence of shedding of the membrane-bound form. sDPP4 level increases in obese subjects and T2DM patients and may participate in increased insulin resistance in these cases [99]. Membrane-bound DPP4 expression is triggered in case of hypoxia as well as its' shedding [100,101].
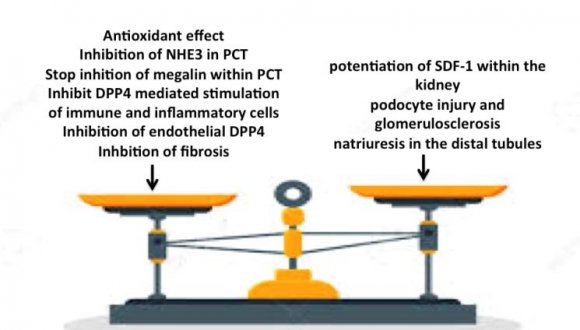
Within the kidney, DPP4 in S1-S3 segments of the proximal convoluted tubules (PCT) are linked to NHE3 and plays a role in salt and water retention through stimulation of this exchanger, NHE3 activity decreases on inhibition of angiotensin II synthesis by captopril [102], or inhibition of DPP4 [103]. Angiotensin II inhibits megalin receptor endocytosis protein expression. This process is reversed by DPP4Is [104]. Treatment with DPP4 inhibitors may reverse reduced uptake of albumin and other low molecular weight proteins by PCT [105]. DPP4 was also discovered on the glomerular endothelium and the base of the foot processes of podocytes [106]. DPP4 is expressed on Tcells, B-cells, macrophages, and dendritic cells in the kidney [98]. Stimulation of DPP4 on the surface of different immune and inflammatory cells may mediate inflammation within the kidney in diabetic patients. DPP4Is decrease inflammation in diabetes. This finding suggests inflammation as an eminent player in DPP4mediated kidney injury [107].
However, in spite of the reduction in urine albumin excretion observed in 3 randomized controlled studies (RCT) in T2DM patients treated with DPP4Is [108][109][110], the only study that specifically looked for the anti-proteinuric effect of linagliptin failed to find a significant impact [111]. Moreover, DPP4Is failed to have a significant impact on doubling of serum creatinine, change in GFR, or ESRD [108][109][110]. On the other hand, administration of linagliptin to T2DM patients that had renal dysfunction and were prescribed ACE inhibitors or ARBs has led to additive significant reduction in albuminuria [112].
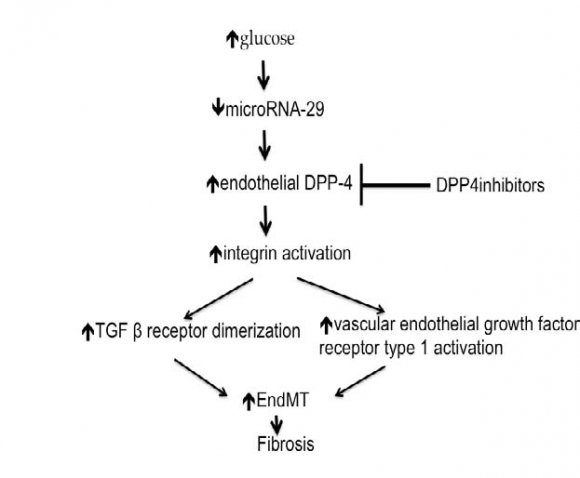
In normoglycemic milieu, microRNA-29 (miR29) suppresses DPP4 gene. In hyperglycemic state, this suppression is lost. As a consequence, cell surface DPP4 activity increases [113]. In diabetic mice, activated endothelial DPP4 induces phosphorylation of adjacent integrin ?1 on the surface of the endothelium. The activated DPP4, together with the phosphorylated integrin ?1 form a complex that up-regulates TGF ? receptor and activates the surface vascular endothelial growth factor receptor type 1(VEGFR1). Up-regulated TGF ? receptor and VEGFR1 stimulate endothelialmesenchymal transition (EndMT) that increases transition to fibroblasts with subsequent increased fibrogenesis [114] [Fig. 6]. However, the lack of significant impact of DPP4Is on GFR in human studies would cast doubts on their favorable effect on renal fibrosis in humans.
The effect of DPP4Is treatment on the retina is debatable. While some investigators reported an increase in retinal endothelial leakage and vascularity [115], others have reported a significant reduction in the risk of diabetic retinopathy progression [116].
The lack of the expected favorable effect of DDP4Is on diabetic microvascular and macrovascular complications of diabetes in spite of the proven molecular and experimental mechanisms can be attributed to potentiation of the stem cell chemokine, stromal cell-derived factor-1 (SDF-1), which promotes inflammation, proliferation and neovascularization [117]. SDF-1 enhances atheromatous plaque growth and instability, cardiac inflammation, and fibrosis [118]. The renal effects of DPP4Is are mainly through potentiation of SDF-1leading to podocyte injury and glomerulosclerosis. SDF-1 also induces natriuresis in the distal tubules, contrary to SGLT2Is and NHE3 inhibitors that act on PCT. Hence, SDF-1 cannot utilize tubuloglomerular feedback to modulate the glomerular hyperfiltration [117,119] [Fig. 7]. SDF-1 may also aggravate both retinopathy and neuropathy [117,120].
7. g) Sodium glucose co-transporters inhibitors
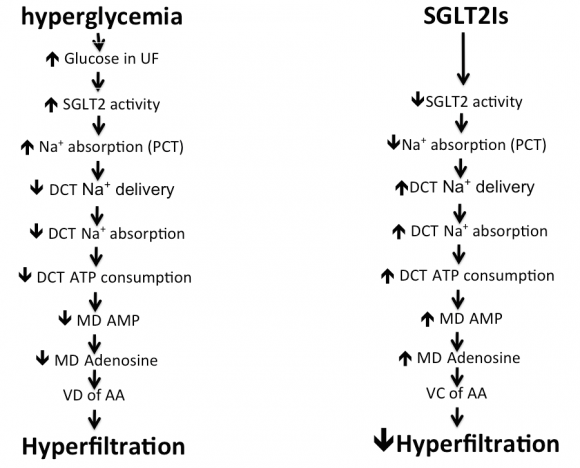
SGLT2Is constitute a recently introduced group that has insulin independent hypoglycemic effect. Three members of this group, namely empagliflozin, canagliflozin, and dapagliflozin are FDA approved and are now used worldwide. By inhibiting the upregulated SGLT2 co-transporters in the brush border of the S1 segment of the PCT, SGLT2Is can reduce the renal threshold for plasma glucose from 196 to 22 mg/dL, thereby enhancing urinary excretion of glucose [121]. They also increase distal sodium delivery and hence distal tubular sodium absorption. Increased adenosine triphosphate (ATP) consumption during sodium absorption with a consequent increase of adenosine production, causes afferent arteriolar vasoconstriction and fall in renal blood flow, reverses hyperfiltration, and accordingly reduces renal injury [Fig. 8]. In addition, SGLT2Is exert other beneficial effects, including reductions in body weight, serum UA, and blood pressure [122]. Excess glucose within the tubular lumen triggers the uric acid transporter GLUT9 within the S3 segment of the PCT and in the collecting duct to excrete UA in exchange with glucose [123]. The antihypertensive effect of SGLT2Is is related to volume depletion, loss of body weight, inhibition of endothelial NHE1 and renal NHE3, and reduction in serum UA [71]. DPP-4= dipeptidyl peptidase; TGF= transforming growth factor; EndMT= endothelial mesenchymal transition.
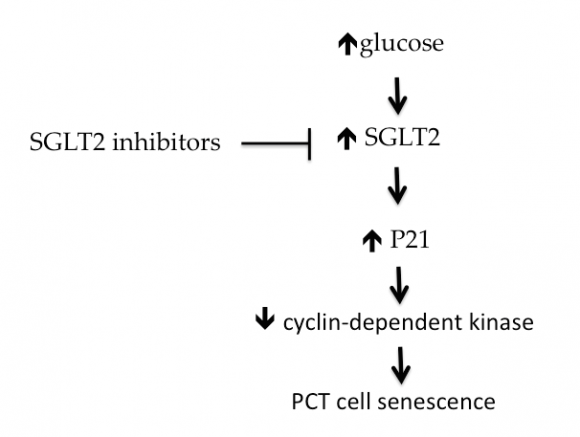
SGLT2Is not only decrease serum UA but they can decrease intracellular fructose metabolism and UA synthesis in the PCT epithelium [124]. Intracellular UA is pro-oxidant. It stimulates NADPH oxidase enzyme activity with consequent increase in production of ROS. This leads to premature senescence of these cells, activation of the renin-angiotensin system, epithelialmesenchymal transition, and activation of the inflammatory cascade through activation of NF-?B [125][126][127] [Fig. 5]. Cyclin-dependent kinase (CDK) inhibits cell senescence. P21 is an inhibitor of CDK and thus promote cell senescence. Hyperglycemia induces P21 while SGLT2Is inhibit this factor within PCT cells [128,129] [Fig. 9]. SGLT2Is also dampen the expression of Toll-like receptor-4, the binding of nuclear DNA for activator protein 1, the increased collagen IV expression as well as the increase in interleukin-6 secretion and interstitial macrophage infiltration induced by hyperglycemia within the renal parenchyma [130]. Moreover, fibrotic and inflammatory genes are suppressed within the diabetic kidney by SGLT2Is [131,132]. Through suppression of intracellular UA production, SGLT2Is inhibits renal gluconeogenesis. Intracellular UA stimulates adenosine monophosphate dehydrogenase (AMPD) enzyme and inhibits adenosine monophosphate kinase (AMPK) enzyme activities. Intracellular AMPD stimulates while AMPK inhibits gluconeogenesis [133]. In healthy personnel, the kidneys participate in endogenous glucose production.
In the fasting state, 20%-25% of endogenous glucose production takes place through renal gluconeogenesis. In T2DM, renal gluconeogenesis increases three fold [134].
Empagliflozin in EMPA-REG trial achieved 55% reduction of the chance of ESRD in T2DM patients with established cardiovascular disease, and an eGFR >30 mL/min/1.73m 2 . The median observation time in EMPA-REG trial was 3.1 years [135]. In comparison, losartan treatment of similar population having DN has led toa 28% delay in the onset of ESRD during a mean follow-up of 3.4 years [136]. Empagliflozin treatment resulted in a 39% reduction of incident or worsening nephropathy, a 38% reduction in progression to overt albuminuria and a 44% reduction in doubling of serum creatinine [137]. The favorable outcome of SGLT2Is is attributable to their effect on glomerular hyperfiltration, blood pressure, body weight, and serum UA in diabetic patients [137][138][139]. SGLT2Is also inhibit NHEs on the surface of cardiomyocytes, endothelial cells,and renal tubular epithelial cells. NHE inhibition can explain the distinguished cardioprotective and renoprotective actions of SGLT2Is [140][141][142]. Decreased renal blood flow induced by SGLT2Is is related to tubuloglomerular feedback and not related to the renin-angiotensin system (RAS) blockade. Empagliflozin and dapagliflozin increase plasma aldosterone and angiotensin II [143,144], together with increased activity of urinary angiotensin converting enzyme and angiotensin converting enzyme2 [145].
2-years treatment of T2DM patients (total of 1450 cases) already kept on metformin with either once-daily canagliflozin 100 mg, canagliflozin 300 mg, or glimepiride titrated to 6-8 mg resulted in eGFR decline by 0.5, 0.9, and 3.3mL/min/1.73m 2 /year respectively (P<0.01 for each canagliflozin group versus glimepiride) in spite of comparable reductions in HbA1c. UAE declined more with canagliflozin 100 mg or canagliflozin 300 mg than with glimepiride. These results further support that the renoprotective effect of SGLT2Is is independent of their glycemic effect [146]. Contrary to DPP4Is and sulfonylurea as that are significantly associated with increased risk of diabetic retinopathy, SGLT2Is were not associated with a higher risk of diabetic retinopathy than placebo among 100 928 patients with T2DM included in 37 independent randomized controlled trials with 1806 diabetic retinopathy events [147]. In the Canagliflozin Cardiovascular Assessment Study (CANVAS), 10 142 T2DM patients were assigned to canagliflozin or placebo. 34% of the patients had ?2 risk factors for cardiovascular events but had no history of previous cardiovascular event (primary prevention cohort), while the remaining 66% had a positive history of cardiovascular event (secondary prevention cohort). The patients were randomly assigned in a ratio of 1:1:1 to either canagliflozin 100 mg, canagliflozin 300 mg or matching placebo. After treatment for a mean of 3.6 years, the primary endpoint (cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke) has occurred less frequently with canagliflozin compared with placebo (26.9 versus 31.5/1000 patient-years; P=0.02). There was no statistical evidence of heterogeneity between the primary and secondary prevention cohorts. Renal outcomes were reduced by 40% and heart failure hospitalization was reduced by 33% in patients treated with canagliflozin [148,149]. 17,160 T2DM patients, including 6,974 with atherosclerotic cardiovascular disease, were assigned for 10 mg Dapagliflozin or placeboin 1:1 ratio and were followed for a median of 4.2 years. Dapagliflozin decreased the composite of cardiovascular death or hospitalizations for heart failure in those with established atherosclerotic cardiovascular disease (ASCVD) and those with only multiple risk factors. The renal composite endpoint (?40% decrease in estimated glomerular filtration rate to <60 mL per minute per 1.73 m 2 of body-surface area, new end-stage renal disease, or death from renal or cardiovascular causes) decreased by 24% in the Dapagliflozin group [150]. When patients with previous myocardial infarction (n = 3,584) were specifically looked for, adverse cardiovascular events were 16% less in the dapagliflozin arm [151]. According to these studies, SGLT2Isshould be prescribed aiming at cardiovascular protection in patients with T2DM and ASCVD [152].
T2DM patients suffering CKD and albuminuria (4400 patients) were randomly assigned to receive canagliflozin100 mg daily or placebo in 1:1 ratio. All the patients had an eGFR of 30 to <90 mL/minute/1.73 m 2 and albuminuria (urine albumin/creatinine ratio>300 to 5000 mg/gm) that were receiving RAS blockers. The primary outcome was a composite of ESRD (dialysis, transplantation, or a sustained eGFR of <15 mL/min/ 1.73 m 2 ), a doubling of the serum creatinine, ordeath from renal or cardio-vascular causes. The projected duration of the study was 5.5 years. Investigators of this study prematurely terminated the trial after a planned interim analysis on the recommendation of the data and safety monitoring committee. This analysis has shown a highly significant reduction of the primary composite endpoint by 34% in patients treated with canagliflozin after 2.6 years of treatment. Patients in the canagliflozin group also had a lower risk of ESRD, hospitalization for HF, and the composite of CV death, myocardial infarction, or stroke. These results indicate that canagliflozin may be an effective treatment for renal and cardiovascular protection in T2DM patients suffering CKD [153]. The observed benefits were obtained mainly in patients whose basal eGFR was between 30 mL/min/1.73 m 2 . In addition, these findings were observed despite very modest differences in blood sugar, weight, and blood pressure between the placebo and the active treatment groups. This suggests that the mechanism of benefit is independent of glycemic control and is likely related to the reduction in single nephron hyperfiltration related to NHE3 inhibition.
The more recent results of the DECLARE -TIMI 58 have supported the favorable effects of SGLT2Is. In this last mentioned trial, 17160 type 2 DM patients were studied using dapagliflozin 10 mg versus placebo in 1:1 ratio for 4.2 years. 47.6% of these patients had GFR >90, 45.1% had GFR between 60 and 90, while only 7.4% of the patients had GFR < 60 ml/min/1.73m 2 . In addition, more than two thirds of the patients had normal urine albumin excretion. Contrary to CREDENCE trial patients where all patients were prescribed RAS blockers, only 81.3% of DECLARE study patients were on RAS blockers. The pre specified composite cardio-Renal end points (sustained decline of at least 40% in eGFR to less than 60 mL/min/1?73m², end-stage renal disease (defined as dialysis for at least 90 days, kidney transplantation, or confirmed sustained eGFR <15mL/min per 1?73 m²), or death from renal or cardiovascular causes were significantly reduced by 24% in the dapagliflozin group while the prespecified composite renal end points (excluding the cardiovascular causes of death from the cardio-renal composite endpoints) decreased by 47%,and the chance to develop ESRD decreased by 56% in the dapagliflozin group. The significant impact of dapagliflozin was encountered in patients having baseline GFR >90, between 60 and 90, in normoalbuminuric patients, in patients with microalbuminuria and in those with overt proteinuria. We would like to emphasize that these favorable effects were only encountered in patients already maintained on either ACE inhibitors or ARBs [154].
8. h) Free oxygen radicals scavengers
Many preclinical studies have overwhelmed the role of reactive oxygen species (ROS) in the pathogenesis of diabetic complications. However, the less favorable outcomes of different antioxidants to prohibit the development or progression of diabetic complications in large clinical trials have dampened the enthusiasm for the use of antioxidant agents in diabetes [155]. Clinical studies using vitamin A, C, and E as antioxidant agents in pre-diabetic and T2DM patients were disappointing. Nuclear factor erythroid 2-related factor 2 (Nrf2) is a transcription factor that protects and restores cell homeostasis upon activation. Although Nrf2 is activated in response to hyperglycemia, this activation does not reach the sufficient level capable to combat the oxidative stress fueled by hyperglycemia [156]. Insufficient Nrf2 activity is often associated with the pathogenesis of diabetes and its complications [157]. Natural products can activate Nrf2, as a potential therapeutic target to control diabetic complications [157,158]. Cruciferous vegetables, grapes, buckwheat, black tea, citrus fruits, apple peels, cinnamon, turmeric, Berberis Mahonia plant, kiwi fruits, the climbing plant Sinomenium acutum, garlic, and Bitter Melon are rich sources of different natural Nrf2 activators [159][160][161][162].
Consumption of 10 gms of broccoli sprouts powder, a rich source of sulforaphane, daily for four weeks was associated with significant improvement in insulin resistance in sixty-three T2DM patients [163]. In a double-blind trial in T2DM patients, the study candidates consumed oral 2×5 mg resveratrol (resveratrol group) or a placebo (control group) for four weeks. Resveratrol significantly decreased insulin resistance, and urine ortho-tyrosine/creatinine ratio as an index of ROS production [164]. A more recent study of ten T2DM subjects, daily consumption of 3 grams of resveratrol for 12 week has increased skeletal muscle Sirtuin1 and adenosine monophosphate kinase enzymes expression. These findings can further support the insulin sensitizing effect of resveratrol [165]. On the other hand, resveratrol supplementation over five weeks in fourteen T2DM patients already kept on diet control did not have a significant effect on glycemic control [166].
In seventy-five patients undergoing primary cardiovascular disease prevention, resveratrol-rich grape supplement significantly decreased highsensitivity C-reactive protein, tumor necrosis factor-?, plasminogen activator inhibitor type 1, and increased anti-inflammatory interleukin-10. The authors concluded that 1-year consumption of a resveratrol-rich grape supplement improved the inflammatory and fibrinolytic activities in high cardiovascular risk and diabetic patients [167]. The beneficial anti-inflammatory effect of resveratrol-rich grape supplement was further supported in a later study of 35 T2DM male patients. One-year consumption of resveratrol-rich grape supplement down-regulated the expression of pro-inflammatory cytokines in circulating mononuclear cells [168]. However, a more recent and larger study failed to prove a significant impact of low (40 mg/day) and high doses (500 mg/day) used for 6 months on fasting blood sugar, glycated hemoglobin or c-reactive protein [169]. When 36 dementia-free, T2DM 49-78 years old patients consumed single doses of synthetic trans-resveratrol (75, 150 and 300 mg) at weekly intervals, trans-cranial Doppler ultrasound both before and 45 min after treatment had shown that only the 75 mg dose was efficacious to improve the cerebral vasodilator responsiveness in both middle and posterior cerebral arteries [170]. In addition, a single 75 mg dose of resveratrol was found to improve neurovascular coupling and cognitive performance in Thirty-six T2DM adults aged 40-80 years [171]. A more recent study has shown that a daily 100mg resveratrol supplementation for twelve weeks in 50 T2DM patients was associated with a significant decrease of arterial stiffness estimated by cardio-ankle vascular index [172].
When the endothelial function was assessed using digital volume plethysmography to measure the changes in the reflective index, oral intake of curcumin 150 mg twice daily for eight weeks has lead to a significant improvement in endothelial function [173]. Supplementation of twenty T2DM patients suffering overt proteinuria with 22mg of curcumin three times daily for two months significantly decreased urinary protein excretion and urine IL-8 beside serum levels of TGF-? and IL-8 [174]. Curcumin 500 mg three times daily was administered for nine months to 120 pre-diabetic patients and significantly improved insulin resistance and beta cell function with consequent prevention of diabetes [175]. Further studies supported the favorable anti-diabetic effect of curcumin [176][177][178].
9. i) Recommendations of diabetes associations
In October 2018, the European Association for the Study of Diabetes (EASD) and the American Diabetes Association (ADA) issued an updated consensus statement on management of hyperglycemia in T2DM patients. This consensus showed-up during the annual meeting of EASD in Berlin, Germany. In this consensus, patients with clinical CV disease should receive one of SGLT2Is or GLP-1RAs, while in patients with CKD or clinical HF and ASCVD, SGLT2Is should be considered [179]. The choice of diabetes therapies as recommended by the American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology (ACE) must be individualized based on many attributes including the risk reduction in heart and kidney disease [180].
10. j) Novel markers of Diabetic complications
Mannose-binding lectin (MBL) is a recognized protein of the innate immune system. It is composed of a lectin (carbohydrate-binding) moiety attached to a collagenous moiety. MBL binds to a wide range of sugars that permits MBL to interact with a wide range of viruses, bacteria, yeasts, fungi, and protozoa containing such sugars within their cell walls or membranes. When bound to its target sugar moiety, MBL can activate the complement system in the classic pathway or in C1-independent manner [181]. MBL is independently associated with HbA1c among diabetic patients [182]. MBL is involved in complement activation within the diabetic kidney [183] and was discovered as a possible independent predictor of DR, DN and other vascular complications in type 1 and type 2 diabetes [184][185][186][187][188][189].
In 297 newly diagnosed T2DM patients, serum fibrinogen was a strong predictor for DN [190]. Serum Adiponectin was proved as a strong predictor of DN in both type 1 and type 2 diabetic patients according to a recent meta-analysis of 13 studies of more than five thousand cases [190].
11. II.
12. Discussion
Diabetic complications pose a massive public health and economic burden. The introduction of GLP1RAs, DPP4Is, and SGLT2Is has revived the hope to effectively prevent or slow down the rate of progression of these complications. These hypoglycemic agents have, in addition, a favorable effect on body weight with less likelihood to experience hypoglycemia. In parallel with their evolving evidence of CV and renal protective effects, ADA recommended SGLT2Is as second-or thirdline antihyperglycemic treatment [191]. The updated consensus statement on management of hyperglycemia in type 2 diabetes issued by EASD and ADA has also recommended the early introduction of SGLT2Is and GLP1RAs to diabetic patients with clinical CV disease and SGLT2Is to patients with CKD or clinical HF and ASCVD. These recommendations were founded on the accumulating evidence of the significant impact of these agents in secondary prevention. The lack of similar significant impact in primary prevention is likely related to the relatively short duration of CVOTs. The most famous primary prevention trial in T2DM patients is UKPDS. It took ten years after the end of this study to get significant differences in acute myocardial infarction and overall mortality between intensive therapy group and the standard of care group [192]. In spite of the significant renoprotective effect of canagliflozin in CREDENCE trial in the whole studied group, patients with eGFR? 60 mL/min/1.73 m 2 and patients with UAE ?1000 mg/gm creatinine failed to get the expected benefit [153]. The planned duration of this study was 5.5 years. However, the study was prematurely terminated according to the observed significant difference in the composite endpoints between the 2 arms in the whole group. Given the known long duration of stage 4 DN, the short duration of this study was not enough for patients recruited with eGFR? 60 mL/min/1.73 m 2 and patients with UAE ?1000 mg/gm creatinine to d evelop enough number of primary endpoints. The rate of decline of GFR in DN patients is around 6mL/min/1.73m 2 [146]. The more recent results of the DECLARE -TIMI 58 did support this view. This last mentioned trial continued for 4.2 years. In this study, the significant impact of dapagliflozin was encountered in patients having baseline GFR >90, between 60 and 90, in normoalbuminuric patients, and in patients with microalbuminuria. These favorable effects in patients of DECLARE study are likely related to the relatively longer duration of follow-up [154]. Taking these results into account and according to the accumulating evidence, more energetic primary preventive studies should be designed. These new studies should select newly diagnosed diabetic patients that have laboratory markers of likelihood to develop diabetic nephropathy later during the future course of their disease. The main obstacle for such studies is the duration needed to have enough number of endpoints for adequate statistical analysis. Such long duration may lend such studies very costly and very exhaustive. Given the safety and noninferiority of SGLT2Is, GLP1RAs, and DPP4Is, and the highlighted beneficial effects of these agents, we suggest a more reproducible approach to manage T2DM patients. In addition to T2DM patients with clinical CV disease, and those with ASCVD, patients with high cardiovascular risk should be prescribed SGLT2Is as second-line hypoglycemic agent after metformin. RAS blockers should be additionally added to guarantee optimum benefit. In the case of morbid obesity, inability to control food avidity or hyperglycemia, GLP1RAs can be used instead of SGLT2Is. In case of failure of SGLT2Is to achieve the glycemic target, either DPP4I or GLP1RA should be added as the third-line agent. SGLT2I can be added on top of GLP1RA if the later failed to achieve the glycemic target. Routine screening of diabetic patients for likelihood to develop diabetic nephropathy using the early predictors like serum MBL, fibrinogen, or adiponectin can help to select patients prone to develop diabetic nephropathy. These patients should be prescribed SGLT2Is to prevent the development of the disease instead of waiting untill they develop albuminuria. This primary prevention approach can completely abort the development of DN instead of the current secondary prevention that just postpones the event for few months or years.
| endothelial dysfunction via calcium-dependent 66. Bartáková V, Kuricová K, Pácal L, Nová Z, | ||
| calpain. Cardiovasc Res. 2008 Nov 1; 80(2): 255-62. 44. Packer M. Activation and Inhibition of Sodium-Hydrogen Exchanger Is a Mechanism That Links the Pathophysiology and Treatment of Diabetes Mellitus with that of Heart Failure. Circulation. 2017 Oct 17; 136(16): 1548-1559. Dvo?áková V, ?vr ?ková M, et al., hyperuricemia: 3-year randomized parallel-45. Katusic Z S, Austin S A. Neurovascular Protective Function of Endothelial Nitric Oxide controlled study.Clin Endocrinol (Oxf). 2015 Oct; -Recent 83(4): 475-82. Advances. Circ J. 2016 Jun 24; 80(7): 1499-503. 46. Alves-Lopes R, Neves K B, A C, Harvey A, Carneiro F S, Touyz R M et al. Internal pudental artery dysfunction in diabetes mellitus is mediated by NOX1-derived ROS-, Nrf2-, and Rho kinase-dependent mechanisms. Hypertension. 2016; 68: 1056-64. 47. Wu S1, Gao X, Yang S, Liu L, Ge B, Yang Q. Protective effects of cariporide on endothelial dysfunction induced by homocysteine. Pharmacology. 2013; 92 (5-6): 303-9. 48. Packer M. Role of the sodium-hydrogen exchanger in mediating the renal effects of drugs commonly used in the treatment of type 2 diabetes. Diabetes Obes Metab. 2017 Dec 11. 68. Kanji T, Hyperuricemia and risk of incident hypertension: a systematic review and meta-analysis of observational studies. PLoS One. 2014 Dec 1; 9(12): e114259. 72. Vilsbøll T, Holst J J. Incretins, insulin secretion and type 2 diabetes mellitus. Diabetologia. 2004 Mar; 47(3): 357-366. 73. Kalra S Glucagon-like peptide-1 receptors agonists (GLP1 RA). J Pak Med Assoc. 2013 Oct; 63(10): 1312-5. 74. Ross S A, Ekoé J M. Incretin agents in type 2 diabetes. Can Fam Physician. 2010 Jul; 56(7): 639-48. 75. | Arterioscler Thromb Vasc Biol. 2011; 32(2): 467-473. 33. Liu J, Jahn L A, Fowler D E, Barrett E J, Cao W, Liu Z. Free fatty acids induce insulin resistance in both cardiac and skeletal muscle microvasculature in humans. J Clin Endocrinol Metab. 2011; 96(2): 438-446. 34. Rask-Madsen C, Li Q, Freund B, Feather D, Abramov R, Wu I H, et al., Loss of insulin signaling in vascular endothelial cells accelerates atherosclerosis in apolipoprotein E null mice. Cell Metab. 2010; 11(5): 379-389. 35. Tremolada G, Del Turco C, Lattanzio R, Maestroni S, Maestroni A, Bandello F, et al., The role of angiogenesis in the development of proliferative diabetic retinopathy: impact of intravitreal anti-VEGF treatment. Exp Diabetes Res. 201210.1155/2012/ 728325 36. Satchell S C. The glomerular endothelium emerges as a key player in diabetic nephropathy. Kidney Int. 2012; 82(9): 949-951. 37. Stirban A Microvascular dysfunction in the context of diabetic neuropathy. Curr Diab Rep. 2014; 14(11): 541. 38. Exalto L G, Whitmer R A, Kappele L J, Biessels G J. An update on type 2 diabetes, vascular dementia and Alzheimer's disease. Exp Gerontol. 2012; 47(11): 858-864. 39. Kanetsuna Y, Takahashi K, Nagata M, Gannon M A, Breyer M D, Harris RC, et al.,. Deficiency of endothelial nitric-oxide synthase confers susceptibility to diabetic nephropathy in nephropathy-resistant inbred mice. Am J Pathol, 2007, 170: 1473-1484. 40. Zhao H J, Wang S, Cheng H, Zhang M Z, Takahashi deficiency produces accelerated nephropathy in diabetic mice. J Am Soc Nephrol, 2006, 17: 2664-2669. 41. Huber J D, Bentzien J, Boyer S J, Burke J, De Lombaert S, Eickmeier C, et al., Identification of a potent sodium hydrogen exchanger isoform 1 (NHE1) inhibitor with a suitable profile for chronic T, Fogo A B, et al., Endothelial nitric oxide synthase | Year 2019 Volume XIX Issue V Version I ( D D D D ) K Global Journal of Medical Research |
| dosing and demonstrated cardio protective effects | ||
| in a preclinical model of myocardial infarction in the | ||
| rat. J Med Chem. 2012 Aug 23; 55(16): 7114-40 | ||
| 42. Sarigianni M, Tsapas A, Mikhailidis D P, Kaloyianni | ||
| M, Koliakos G, Fliegel L, et al., Na+ H+ | ||
| exchanger-1: a link with atherogenesis? Expert Opin | ||
| Investig Drugs. 2010 Dec; 19(12): 1545-56. | ||
| 43. Wang S, Peng Q, Zhang J, Liu L. Na+/H+ | ||
| exchanger is required for hyperglycaemia-induced | ||
| © 2019 Global Journals |