1. Introduction
robiotic bacteria, which are traditionally studied to aid intestinal functions, are also currently being researched for benefits in other parts of the body such as the oral cavity. Thus, commensal microorganisms of the mouth, ears, and nose have been researched for the prevention or treatment of oropharyngeal diseases such as periodontitis, caries, sore throat, and otitis.
This study aimed to evaluate the presence or absence of toxicity of the Streptococcus salivarius subsp salivarius BIO5 strain 1 .
The Streptococcus salivarius subsp salivarius species is an indigenous microorganism of the mouth, and its niches are the tongue and mucous membranes. It is a bacterium that sets in soon after birth, remaining dominant from childhood through life 2,3 . Some strains are capable of producing bacteriocin-like inhibitory substances (BLIS) 4 in vitro against the agent of bacterial tonsillitis -Streptococcus pyogenes or Group A Streptococcus (GAS). Tonsillitis is one of the most common childhood diseases, which when repeated may lead to the development of rheumatic fever and cause severe sequels 5 . Considering this knowledge, the possibility of performing replacement therapy was considered by introducing positive bacteriocin strains through some vehicle into the oral cavity of children with tonsillitis. The purpose of performing toxicity tests with an indigenous microorganism of the oral cavity is that this species -Streptococcus salivarius subsp salivariushas been studied as the ideal probiotic for controlling bacterial tonsillitis caused by Streptococcus pyogenes. Despite being a beneficial and non-pathogenic saprophytic microorganism, it is necessary to confirm that the strain in question is completely safe. As a probiotic, S. salivarius has been used with the K12 strain successfully through oral tablets preceded by a mouthwash with an antiseptic 6,7,8 .
The BIO5 strain of Streptococcus salivarius has been studied for the development of a product other than the K12 strain that is administered through tablets. Lately, new strains of S. salivarius have been studied for the same purpose: control and treatment of tonsillitis as well as otitis and halitosis 9,10 .
Therefore, confirmatory tests for the complete absence of toxicity of the Streptococcus salivarius BIO5 strain are required.
2. II.
3. Material & Methods
We obtained one strain with clear evidence of bacteriocin-like production and inhibition against most of the 100 strains of S. pyogenes. The Streptococcus salivarius subsp salivarius BIO5 strain was tested by PCR11 to identify S. salivarius.PCR primers specific for S. salivarius, Ssa442F (5´-AAC GTT GAC CTT ACG CTA GC-3´) and Ssa2712R (5´-GAT TCT GTC AAA GAA GCC AC-3´) were used to amplify a 2271 bp fragment from dextranase (dex) gene. Isolates were prepared for PCR by pelleting 3 ml of bacteria grown in BHI broth, subsequently suspended in 1 ml of sterile MilliQ water. 5 µl aliquots of the cell suspension were used in a 50 µl reaction containing Reaction Buffer Biotools 1x with 2 mM MgCl2 (Madrid, Spain), 1 µM of each primer (Invitrogen, Carlsbad, CA, USA), 2 mMdNTPs mix (Invitrogen) and 2, 0 U of Taq polymerase (Invitrogen). The amplification reaction was performed in an Eppendorf Mastercycler gradient thermal cycler (Eppendorf, Hamburg, Germany) as described byIgarashi et al11 as it follows: 95°C for 10 min followed by 26 cycles of denaturation at 94°C for 1 min, annealing at 55°C for 1 min and extension at 72°C for 1 min. The last cycle comprised denaturation at 94°C for 1 min, annealing at 55°C for 1 min and final extension step at 72°C for 5 min. The PCR fragments were subjected to electrophoresis on 1.5% agarose gel in TAE buffer 1x and stained with ethidium bromide.
4. Toxicity tests
The toxicity tests for this study were clinical, ophthalmic lesions, behavioral observations, mortality, anatomopathological examination, biopsy, hematology, serum biochemistry, and urine analysis 12 . The authors complied with the guidelines for the care and use of laboratory animals as described by the U.S. National Institutes of Health.
5. a) Test Product Preparation
The BIO5 strain was seeded from blood agar stock on Mitis Salivarius agar and incubated at 37°C/24h for confirmation of culture purity. From this growth, suspensions were prepared in saline solution at concentrations from 10 8 to 10 9 CFU/ml. This preparation was called Test Product.
6. b) Animal Studies
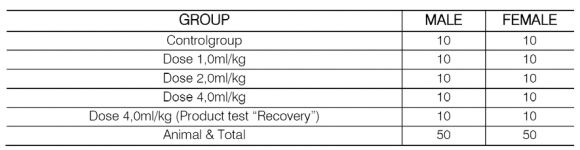
Animals of the Rattusnovergicus species, Wistar Lineage, albino rats were used. A total of 100 animals were initially used -50 males and 50 females. These animals received three different doses of the Test Product for a period of 28 consecutive days, namely 1.0, 2.0, and 4.0 ml/kg. The animals originated from the breeding room of the company CIALLYX Laboratórios & Consultorias-Paulínia, São Paulo, Brazil.
? Administration in repeated doses, in a rodent (rat albinus "Wistar"), of the test product (1.0, 2.0 and 4.0 ml) by kg of body weight of 10 animals per dose (total of 50 males and 50 females), which received each of the dosages of the test product plus a control group and a "recovery group". ? Clinical, visual, and behavioral observations were made over the entire period of the treatment of the animals with different doses of the test product, i.e., for 28 consecutive days. There were two daily observations: in the morning and the afternoon. ? Samples of blood/plasma (with the help of a syringe moistened with heparin or not) were collected for additional tests including complete hematology, serum biochemistry tests, electrolytes, and urine analysis.
III.
7. Results
8. a) Experimental Design
? Mortality No deaths were detected throughout the acclimation phase and experimentation (Table 1).
9. ? Anatomopathological examination
All animals that survived the various treatments after 28 days of observation were deeply anesthetized and euthanized ethically. The procedure was necropsy on all rats. Potential injuries were recorded and tissue was collected for histopathological examinations.
10. ? Biopsy
No lesions were found in organic and/or any substantial tissue of animals examined by necropsy (adrenal, aorta, articulation, spleen, bladder, brain, cerebellum and pons, heart, esophagus, stomach, pylorus and cardio, epididymis, salivary gland, liver, testicle, duodenum, ileum, jejunum, cecum, colon, rectum, larynx, lymph nodes, bone marrow, skeletal muscle, prostate, thyroid, pancreas, skin, lungs, kidneys, thymus, thyroid, and trachea.
11. ? Test substance administration
No problems related to the Test Product administration were found. ? Changing the standard of feed and water consumption No changes were observed in the patterns of water and feed intake, both in the acclimation phase (day zero to day seven) and throughout the experimental period (from day zero to day 28).
12. ? Daily observations
No changes were detected with behavioral and/or clinical significance during the entire acclimation phase and experimentation.
13. ? Ophthalmology
There were no problems and/or specific ophthalmic lesions in any of the animals treated throughout the acclimation phase and/or experimentation.
14. ? Hematology
Different hematological parameters were analyzed in animals treated with repeated doses of the test product observed for 53 consecutive days, and no changes were verified (Table 2).
15. ? Serum biochemistry
Different parameters of serum biochemistry were analyzed in animals treated with repeated doses of the test product observed for 53 consecutive days, and no changes were verified (Table 3).
16. ? Urine analysis
Different parameters of urine were analyzed in animals treated with repeated doses of the test product observed for 53 consecutive days (Table 4). Urine analysis (quantitative method) of 10 animals (males) per group receiving doses of 1.0, 2.0, and 4.0 ml/kg. Averages or rankings for the following parameters: color, turbidity, pH, presence of leukocytes (cells/ml),presence of glucose (mg/dL), bilirubin, and presence of blood (erythrocytes/ml).
17. Discussion
All articles published on the Streptococcus salivarius subsp salivarius species agree that this microorganism is an indigenous and saprophyte inhabitant of the human oral cavity. It is installed on the tongue and mucous membranes shortly after birth, remaining in these habitats for life 3,4 . It is an abundant and predominant isolate of the human oral microbiota with the basic function of protecting this environment by hindering or preventing the entry of other foreign or pathogenic microorganisms, aiding the control of the ecological balance of the microbiota and oral health.
In recent years, several authors have been studied the Streptococcus salivarius K12 strain as a probiotic in the treatment of tonsillitis caused by S. pyogenes, showing promising results 6,7,8 .
As the S. salivarius species has only recently been used as a probiotic, when searching the literature for toxicity data of this species we found rare reports of toxicity studies, because it is a saprophytic and indigenous microorganism of the oral cavity. The only study on S. salivarius toxicity was carried out with the K12 strain, in which the authors analyzed toxicity, antibiotic resistance, virulence determinants, the production of deleterious metabolic by-products, and genetic stability 13 . The various sets of data obtained in this study showed no evidence of toxicity and no acute or subacute toxicity effects associated with the K12 strain, leading to the cure of tonsillitis and otitis.
La Mantia et al 9 used the S. salivarius 24SMB and Streptococcus oralis 89a nasal spray Rinogermina for preventing recurrent acute otitis media in children. The results showed that all actively treated children with the highest acute otitis had a reduction of recurrence, whereas only 50% of the children in the control group had reduced the disease.
Marquart et al 14 studied virulence aspects of 22 streptococci strains isolated from endophthalmitis, one of which was Streptococcus salivarius. This strain showed amikacin resistance, vancomycin sensitivity, and intermediate ceftazidime sensitivity. Bacterial genomic DNA from each of the 22 isolates was tested for the presence of the gene encoding pneumolysin, ply. This base pair gene is present in S. pneumoniae and encodes a cytotoxin involved in virulence, but the S. salivarius strain did not present it. The protease activity was also negative for this strain, as well as cytotoxicity or hemolytic.
To identify putative probiotics, Frick et al 15 tested some commensal bacteria on their toxicity, invasiveness, inhibition of Yersinia-induced inflammation in vitro and in vivo, and modulation of dextran sodium sulfate (DSS)-induced colitis in mice. None of the commensal bacteria tested, including Streptococcus salivarius, was toxic for or invaded the epithelial cells.
As the authors cited, despite different studies, our results also showed the absence of any virulent activity in vivo and in vitro, proving that the S. salivarius BIO5 strain can be used as a probiotic in the treatment or prevention of bacterial tonsillitis or other infections in humans.
The K12 strain is administered through tablets. Beforeits administration, a mouthwash with strong antiseptics is required to eliminate part of the microbiota and facilitate the introduction of the strain in question. The authors who used S. salivarius 24SMB strains used an intranasal administration 9,10 .
As we are discussing new bacteria and proposing their use as probiotics, it is appropriate to refer to the Food and Agriculture Organization (FAO), World Health Organization (WHO), and the International Scientific Association for Probiotics and Prebiotics (ISAPP).
In 2001, the Food and Agriculture Organization of the United Nations and the World Health Organization 16 debated the field of probiotics, starting with the definition: "live microorganisms, which when administered in adequate amounts confer a health benefit on the host".
Revisiting the term 'probiotic' in October 2013 by the International Scientific Association for Probiotics and Prebiotics (ISAPP) 17 to discuss the field of probiotics, a more grammatically correct definition would be worded as, "live microorganisms that, when administered in adequate amounts, confer a health benefit on the host". The differentiation between probiotic and commensal was also established. Although probiotics are isolated from normal microbiota, as long as they are not isolated and proven to be beneficial to health, they cannot be called probiotics.
As the literature already shows several works published successfully on the treatment of tonsillitis, especially in children, with S. salivarius, we aim to use the BIO5 strain in the treatment and prevention of this disease but using a different vehicle from those used so far.
Confirming the absence of toxicity of the Streptococcus salivarius subsp salivarius BIO5 strain, we De Grandi et al 10 studied the efficiency of nasal instillation of S. salivarius 24SMBc and S. oralis 89a. In particular, nasal swabs were sampled one, two, and four weeks after seven days of treatment with Rinogermina. They analyzed modulations of the abundance of pathogenic species such as Corynebacterium diphtheriae, Haemophilus parainfluenzae, Moraxella catarrhalis, Prevotella denticola, Prevotella melaninogenica, Rothia dentocariosa, Staphylococcus aureus, and Streptococcus pseudopneumoniae. They characterized a significant temporary decrease in those species. The beneficial effects of S. salivarius 24SMBc and S. oralis89a nasal intake were assessed but seemed to be restricted in specific temporal windows. feel confident in performing the next step, i.e. the study in humans, with this new strain, hoping we can call it a probiotic.
V.
18. Conclusion
The test fulfilled the purposes to evidence potential toxic effects on animals -males and femalesin any of the doses (1.0, 2.0, 4.0 ml/kg). Neither hematological nor pathological changes were observed in serum biochemistry among many different groups. The results obtained allowed us to affirm that the test product did not induce toxic effects evident in doses used of the Test Product analyzed, which is Streptococcus salivarius subsp salivarius BIO5. Therefore, the Streptococcus salivarius BIO5 strain can be safely used in the prevention or treatment of bacterial tonsillitis in humans as it has been shown that such strain did not cause any toxic effects in animals.
| Group | Product Dose | Animal number | Sex Number Death | Mortality % | |
| 1 | Product Test / 1,0 ml/kg | 10 | M | 0 | 0 |
| 2 | Product Test / 2,0 ml/kg | 10 | M | 0 | 0 |
| 3 | Product Test / 4,0 ml/kg | 10 | M | 0 | 0 |
| 4/"Recovery" Product Test / 4,0 ml/kg | 10 | M | 0 | 0 | |
| 1 | Product Test / 1,0 ml/kg | 10 | F | 0 | 0 |
| 2 | Product Test / 2,0 ml/kg | 10 | F | 0 | 0 |
| 3 | Product Test / 4,0 ml/kg | 10 | F | 0 | 0 |
| 4/"Recovery" Product Test / 4,0 ml/kg | 10 | F | 0 | 0 | |
| Low set | Intermed | Highest | Normal | |||
| Male | Control | Dose | Dose | Dose | Values | Unit |
| ml/kg | ml/kg | ml/kg | Rats A | |||
| Total number erythrocytes | 7,6 | 7,2 | 7,7 | 8,1 | 6,0-9,0 | 10 12 /L |
| Hemoglobinconcentration | 14,1 | 14,7 | 14,9 | 15,4 | 11,0-17,0 | g/dL |
| Hematocrites | 44,3 | 42,4 | 41,5 | 42,0 | 38,0-50,0 | % |
| MCHC | 31,9 | 31,7 | 32,6 | 32,9 | 31,0-36,0 | g/dL |
| % reticulocites | 0 | 0 | 1 | 1 | 1,0-4,0 | % RBC |
| Total number granulocytes | 4,7 | 4,4 | 4,9 | 5,1 | 1,0-6,0 | 10 9 /L |
| % de granulocytes | 16,8 | 17,8 | 18,9 | 22,2 | 5,0-30,0 | % |
| Numberlymph /monoc | 7,5 | 7,3 | 6,6 | 5,9 | 4,0-16,0 | 10 9 /L |
| Total leukocytes | 12,8 | 12,2 | 11,6 | 13,7 | 4,0-17,0 | 10 9 /L |
| Numberlymphocytes | 8,3 | 9,6 | 9,2 | 9,2 | 3,0-15,0 | 10 9 /L |
| Numberneutrophils | 3,7 | 4,1 | 5,1 | 5,2 | 0,0-6,0 | 10 9 /L |
| Numbermonocytes | 1,0 | 1,4 | 1,1 | 1,5 | 0,2-2,0 | 10 9 /L |
| Numbereosinophils | 0,1 | 0,2 | 0,4 | 0,2 | 0,0-0,5 | 10 9 /L |
| Numberbasophils | 0 | 0 | 0 | 0 | Raros | 10 9 /L |
| Numberplatelets | 756 | 657 | 678 | 458 | 800-1400 | 10 9 /L |
| Coagulation time | 194 | 192 | 224 | 324 | 100-500 | s |
| Carrier | Dose ml/kg | Dose ml/kg | Dose ml/kg | Values Rats (A) | Units | |
| ligth | ligth | ligth | ligth | ligth | ||
| Color | yellow | yellow | yellow | yellow | yellow | |
| transluce | transluce | transluce | transluce | transluce | ||
| pH | 7,5 | 7,1 | 7,7 | 7,1 | 6,0 -8,0 | |
| Leukocytes | 25 | 25 | 0 | 0 | until 100 | Leucocytes/ ml |
| Protein | 30 | 30 | 0 | 0 | until 100 | mg/dl |
| Glucose.. | Negative | Negative | Negative | Negative | Negative | Negative |
| Ceton | 0 | 15 | 0 | 15 | until 15 | mg/dl |
| Urobilinogen | Normal | Normal | Normal | Normal | Normal | mg/dl |
| Bilirrubin | Negative | Negative | Negative | Negative | Negative | |
| Blood. | Negative | Negative | Negative | Negative | Negative | erythrocytes /ml |
| Animal clinical chemistry/A pratical guide for toxicologists and biomedical researchers. G.O.Evans / CRC Press | ||||||
| 0 or 4.0 |