1.
products [1]. And as a result, food consumption was the major source for aluminum intake under physiological conditions [3]. The widespread presence of aluminum, both in the environment and in food, made it almost impossible to avoid exposure to this metal ion [4]. Aluminum -containing diet was mainly corn, yellow cheese, salts, herbs, spices, and tea, [5]. It entered in production of cosmetics such as antiperspirant and deodorant preparation [6]. Moreover, it incorporated in some medications such as antacids, buffered aspirin, antidiarrheal products, vaccine, and allergen injection [7], [8], and [9]. Also, it used as a component of veterinary medicine, glues, and disinfectants [10]. Aluminum sulfate added as a coagulant agent during the purification process of drinking water [11]. Recently, Aluminum in drinking water led to the development of many diseases and health problems [1]. Aluminum chloride was suggested to induced reproductive toxicity and exerted a significant adverse effect on steroidogenesis [12]. Aluminum compound had serious effects on energy metabolism and hematology [13], [14]. It enhanced free radicals, and changed the enzymatic activities [15], with emphasis on the alterations in the metabolism of testis, epididymis and vas deferens that led to poor sperm motility and reduction in fertility rate in mice [16], [17]. It reduced fertility and reproductive activity [18], [19], and also affected the process of development of embryos by its embryotoxicity and teratogenicity [20]. Liobet et al. [21], Khattab [22], Guo et al. [23], and Fairoze [24]; observed histological changes including necrosis of spermatocytes and spermatids in the testis of male mice treated with aluminum nitrate, and degeneration in the structure of spermatogenesis and formation of giant cells in mice testis treated with aluminum chloride, severe damage within seminiferous tubules and vascular degeneration on spermatogenic and Sertoli cells cytoplasm.
Propolis was a natural resinous mixture produced by honeybees from substances collected from parts of plants, buds, and exudates. Due to its waxy nature and mechanical properties, bees used propolis in the construction and repair of their hives, sealing openings, and cracks, smoothing out the internal walls, and as a protective barrier against external invaders like snakes, lizards, winds and rains increasing the amounts of aluminum in the ecosystems. This element accumulated in plants and water. Potentially increased its harmful effect; particularly, it induced free radicalmediated cytotoxicity and reproductive toxicity. Propolis was a resinous material collected by bees from bud and exudates of the plants, mixed with bee enzymes, pollen, and wax. Due to its waxy nature and mechanical properties, bees used propolis in the construction and repair of their hives, and as a protective barrier against external invaders, or weathering threats, Current antimicrobial properties of propolis, was helping for wound healing, treatment of burns, herpes simplex and genital herpes. The present work studied the protective effect of propolis against the reproductive toxicity of aluminum chloride in male rats. Material and Methods: Sixty male Wistar Albino rats (average weight 200 g) were used and divided into three groups; each contained twenty rats. Groupe I, was a control group, group II, was treated with aluminum chloride (80 mg / Kg. body weight) through an orogastric tube, daily for six weeks. Group III, was given aluminum chloride (80 mg / Kg. body weight), concomitant with (200 mg / Kg. body weight) ethanol extract of propolis, for the same root and period.
Results: Study of the second group revealed degeneration and necrosis of the seminiferous tubules, as demonstrated with the light microscope. The ultrastructure study revealed dilated irregular tubular membrane, Sertoli cell, spermatogonia, and early spermatid with vacuolated cytoplasm and irregular nuclei, swollen mitochondria, dilated irregular tubular membrane. Apparent improve in the light and ultrastructure study of the third group. The results concluded the administration of aluminum chloride to adult male rats would cause adverse effects on reproductive efficiency that could be overcome by the protective effect of propolis.
2. I. Introduction
luminum was widely spread in our environment. It was the third most abundant element on the earth's crust [1]. It has no known biological role in living organisms and may be classified as toxic metal [2]. It was widely used for the manufacturing of many A [25], [26]. It possessed a characteristic and pleasant aromatic smell and various colors from transparent propolis, yellow, green, red, or dark brown, depending on the site of origin, source, and age of the resins, it was hard and brittle, and became soft, pliable, gummy, and very sticky when heated [27]. Raw propolis was composed of around 50% resins, 30% waxes,10% essential oils, 5% pollen, and 5% of various organic compounds [28], [29], and [30]. Propolis was soft, pliable, and sticky substance at 25?C to 45?C, above 45?C; it became gummy, and liquid at 60?C to 70?C, but for some samples the melting point might be as high as100?C [27]. Propolis was extracted with a suitable solvent, the most common solvents used for extraction were water, ethanol, methanol, chloroform, ether, and acetone [31]. According to Ugur and Arslan [32], the use of different solvents changed the biological activity of the main active constituent in propolis. The aqueous extract of propolis showed higher antioxidant activity compared to the ethanol extract of propolis; this was due to its higher polyphenols content. So, it used in prevention of various free radical related diseases. Ethanolic extracts of propolis showed high antibacterial activity against Gram-positive, but least activity against Gram-negative bacteria [33], [34]. Selvan et al. [35] reported that bee propolis, in combination with chlorhexidine, possessed high antimicrobial activity against streptococcus mutants. Propolis was the bee product with the highest antifungal activity as tested with 40 yeast strains of C. Albicans [36], [37]. Propolis showed fungicide effects that prevented juice spoilage [38]. According to Dota et al. [39]. The propolis extract had fungicidal effect against clinical yeas, and also for of the vulvovaginal candidiasis. The European propolis cured some human diseases caused by protozoa [40], [41] and [42]. Propolis was known for its antioxidant properties [43], [44]. The antioxidants present in propolis played a great role in its immunomodulatory properties [45]. All propolis had strong antioxidant activity due to their contents of phenols and flavonoids which increased the cellular immune response [46]. Active flavonoids and cinnamic acid derivatives were responsible for the anti-inflammatory activity of propolis [47], [48], [49], and [50]. Propolis provided beneficial effect on human health, due to its antimicrobial, antiviral, and antioxidant properties, it was widely used in human and veterinary medicine. It entered in manufacturing of cosmetics, medication for cold syndrome, and flu-like infections, treatment of wounds, burns, and acne. It was commercially available in the form of capsules, creams, mouthwash solutions, toothpaste, and, throat lozenges [27].
3. II. Material and Method
Chemicals: Aluminum chloride (AlCl3) came from Guangdong Guanghua Sci-Tech Co. Ltd., Shantou, Guangdong, China, and manufactured by Yueyang Jiazhiyuan Biological Co. Ltd., China.
The dose of aluminum chloride was 80 mg/kg. orally, daily, for six weeks [51].
An ethanol extract of propolis came from Boiron et Cie, Lyon, France.
The dose of the ethanol extract of propolis was 200 mg/kg. orally, daily, for six weeks [52].
Experimental design: Sixty male rats of proven fertility divided into three groups.
Group I: Control group, rats received 2.0 ml. distilled water orally, daily, for six weeks.
Group II: Treated group, rats received aluminum chloride 80 mg / Kg. Orally, daily, for six weeks, it was 6 mg. Dissolved in 1.0 ml. Distilled water. Group III: Treated group, rats received aluminum chloride as the same dose, root and period for group II, and 200 mg/kg. Ethanol extract of propolis, orally, daily, for six weeks, it was about 40 mg. Dissolved in 1.0 ml. Distilled water. After the designed period, the testes were dissected out, trimmed off the attached tissues. Specimens of the testes fixed immediately in 10% buffered formalin for histological study.
4. For Histopathological and Ultrastructural Examination:
According to Bancroft and Gamble [53], tissues were taken from testes of rats were cut and immediately fixed into 10% formaldehyde saline. Tissues were processed for the preparation of paraffin blocks (paraffin method). Sections were cut by rotatory microtome and mounted on glass slides. The sections stained by conventional Hematoxylin and Eosin (H&E), PAS, and Mallory stain, and examined by light microscope.
For the ultrastructural examination: An adjacent section of testis was removed and placed in 0.1M cacodylate buffer containing 3% glutaraldehyde for electron microscopy. After fixation, sections were dehydrated in a series of ethanol rinses, cleared with propylene oxide, and embedded in Epon. (Epon or Epon-Araldite mixtures are the most widely used resins for electron microscopy.) The sections were evaluated using a transmission electron microscope (Joel 1200 Ex-II). [53].
5. III. Results
6. I Histological study
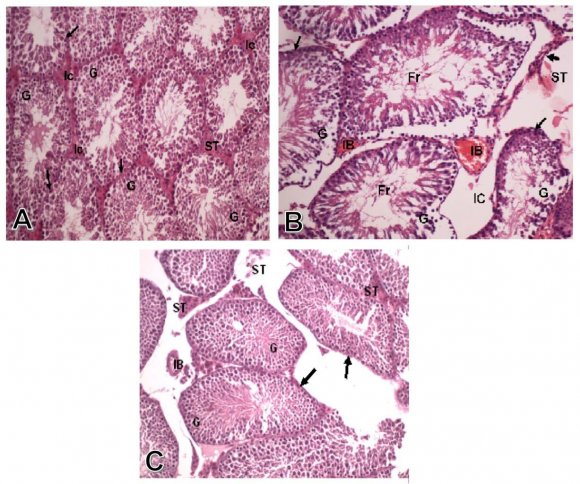
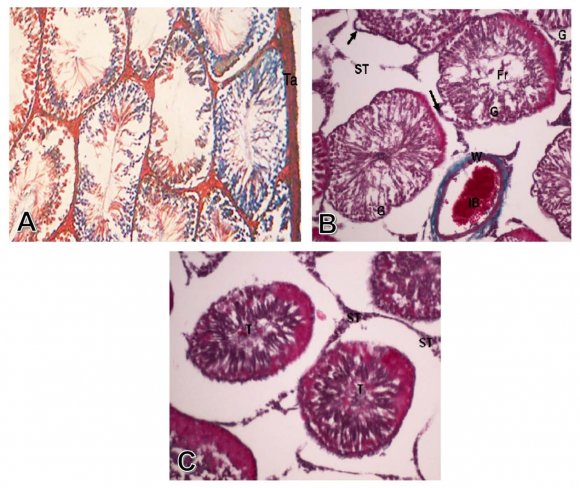
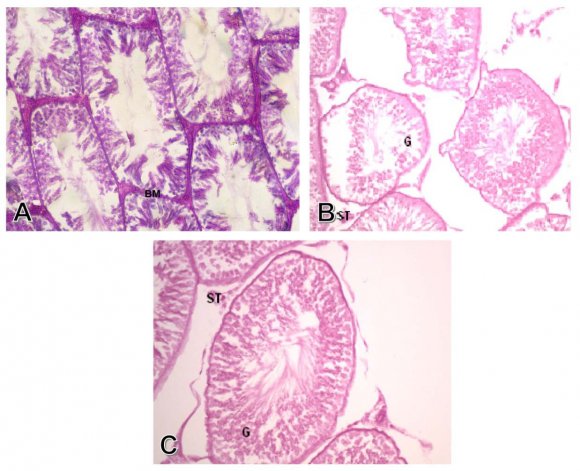
Group I: Control group. The seminiferous tubules were well circumscribed with a clear basement membrane. The germ cells arranged in multiple layers with the sperms occupied the lumen. The interstitial tissue filled the spaces between the seminiferous tubules, which contained blood vessels, and the interstitial cells of Leydig, which appeared in groups of polyhedral cells (Fig. 1-A). The Mallory stain showed the red interstitial tissue and the normal thickness of the tunica albuginea (Fig. 2-A). The PAS stain showed normal thickness and regular pattern of the basement membrane (Fig. 3-A).
7. (arrows), as well as normal interstitial cells of Leydig (ST) and no congestion of interstitial blood vessels (IB). (H&E X250)
Group II: Treated with aluminum chloride. Some of the seminiferous tubules showed marked degeneration and necrosis of the germ cells lining of the tubules. The degenerated tubules showed thick basement membrane and fragments of germ cells in the center of the tubules. There was also edema between the interstitial cells and congestion of interstitial blood vessel (Fig. 1-B). There was some fibrosis of the interstitial tissue which appeared blue with Mallory stain (Fig. 2-B). There was a decreased PAS reaction in the markedly degenerated germ cells lining the seminiferous tubules, and the interstitial cells (Fig 3-B)
8. FIG. 2: (A) A photomicrograph of a transverse section of a control rat testis showing normal thickness of tunica albuginea (Ta).(Mallory X250) (B) Photomicrograph of transverse section of testis of the treated group with alumimium chloride, showing congestion of interstitial blood vessel (IB) with necrotic wall (W), as well as degeneration and necrosis of germ cells (G) lining the seminiferous tubules with fragments of germ cells(Fr) and thickening of tubules basement membranes (arrows).(Mallory X250) (C) Photomicrograph of transverse section of testis of The group treated with alumimium chloride, and propolis, showing normal histological pattern of seminiferous tubules (T) and normal interstitial cells (ST). (Mallory X250).
Group III: Treated with propolis with concomitant administration of aluminum chloride.
The testis of rats treated with aluminum chloride and propolis extract, showed normal histological pattern of the germ cells, basement membrane, and interstitial cells of Leydig, with lack of congestion of interstitial blood vessels (Fig. 1-C).The Mallory stain showed normal histological pattern of seminiferous tubules and the interstitial cells (Fig. 2-C). There were marked increase in PAS reaction of the seminiferous tubules components, and the interstitial cells of Leydig (Fig. 3-C).
9. II Ultrastructure studies
Group I: Control group.
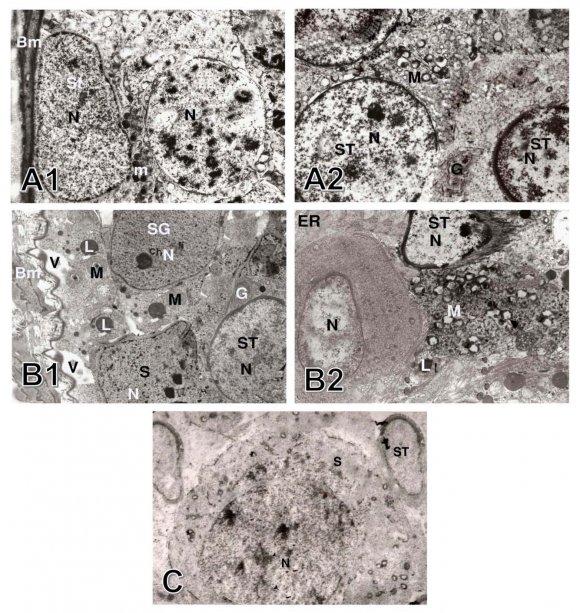
The spermatogonia were large diploid cells that lied against the boundary tissue of the seminiferous tubules. The A-type spermatogonia had large pale ovoid nuclei, a scantly granular cytoplasm, and rough endoplasmic reticulum, the mitochondria were spherical or ovoid, and a simple Golgi apparatus. The B-type cells had rounded nuclei, and a granular cytoplasm. Primary spermatocytes had spherical nuclei with fine granular nucleoplasm, scanty cytoplasm, and ovoid mitochondria that aggregated in groups, the Golgi apparatus arranged in few clusters accumulated at one pole of the cell. The secondary spermatocytes were rarely seen, they entered into the second meiotic division producing the spermatids. The early spermatids were rounded cells with large spherical nuclei that contained clumps of chromatin in a lightly stained cytoplasm, the endoplasmic reticulum had flattened vesicles, the mitochondria aggregated at the periphery of the plasma membrane. The cytoplasm of Sertoli cells extended from the basal lamina to the lumen of the seminiferous tubules. The nucleus was enfolded and the cytoplasm contained abundant endoplasmic reticulum, ovoid Golgi apparatus and spherical mitochondria (Figs. 4-A1 & A2).
Group II: Treated with aluminum chloride.
The seminiferous tubules, showed cellular alteration of cells than those in the control groups; the tubular membranes were dilated with irregular Sertoli cells, the spermatogonia contained vacuolated cytoplasm and irregular nuclei, the mitochondria were swollen, the Golgi apparatus contained lipid droplets. The spermatid cells were atrophied, with degenerated nuclei and defected acrosomal caps, degenerated mitochondria, and dilated endoplasmic reticulum (Figs. Group III: Treated with propolis with concomitant administration of aluminum chloride.
10. 4-B1 & B2).
Sections showed no histological changes in the germ cells which, lined the seminiferous tubules with normal basement membrane (Fig. 4-C).
11. IV. Discussion
Our studies postulated that aluminum chloride exposure caused marked alterations in the histological pattern of the tests which were represented by some of the seminiferous tubules were degenerated, while others appeared necrotic. In addition to degeneration and necrosis of germ cells lining of the seminiferous tubules with thick basement membrane and fragments of germ cells in the center of seminiferous tubules. These results parallel to Liobet et al. [21], Guo et al. [23], Fairoze [24], Burdock [25], Yousef et al. [54], and Hala et al. [55], who founded that aluminum chloride caused histopathological lesion in testis as congestion of interstitial blood vessel, marked degeneration and necrosis of the germ cells, absence of spermatic bundles in seminiferous tubules lumen and this might result from degeneration of the spermatogonia which affected spermatogenesis and maturation of sperms, and this supported our results. The role of aluminum chloride in induction of oxidative stress, this toxic effect might result from its ability to bind to cellular DNA, RNA, and proteins, leading to a decrease of their levels in different tissues [56]. And also, the mechanisms that might explain, the degeneration, the coagulative necrosis, and the epithelial sloughing in our results [56], and [57]. According to Fraga et al. [58] and Kumar et al. [59], the presence of interstitial edema in our results might result from the oxidative damage of capillaries endothelial cells through interference with their membranes, which increased their permeability, the increase in permeability and edema might also occur in inflammatory responses due to the liberation of inflammatory mediators from the degenerated and the necrotic parenchymatous cells, and this might explain inflammatory cells infiltration within testicular parenchyma in our results. Our results showed some fibrosis of the interstitial tissue in the markedly degenerated germ cells lining the seminiferous tubules, as well as decreased PAS reaction of the interstitial cells these findings were in agreement with the results of Chinoy et al. [60] who reported profound disorders in carbohydrate metabolism, follow aluminum chloride toxicity. The degeneration and atrophy of spermatid cells, revealed in the present study agreed with the results obtained by Libet et al. [21] and Guo et al. [23]; who found that there was necrosis in the spermatocytes and spermatids after administration of aluminum nitrite to mice, and the gonadal toxicity of rats exposed to, aluminum chloride, which recorded by Alfrey [61], Krasovskii et al. [62], and Alfrey et al. [63]. In the present study, the pathological changes demonstrated in testicular ultrastructure as irregular Sertoli cell, vacuolated cytoplasm and defected spermatogonia after administration of aluminum chloride were in agreement with Khattab [22], and Kamboji and Kar [64], who reported that, the seminiferous tubules were shrunken with spermatogenic arrest at the primary spermatocytes or spermatogonia stages with the treatment by aluminum sulfate. In the present study, some pathological changes in early spermatid with vacuolated cytoplasm and irregular nuclei after administration of aluminum chloride were in agreement with the work of Mayyas, et al. [65], who reported destruction of the seminiferous tubules with large necrotic areas and degenerated cells. Our present study showed no histological changes in the germ cells which line the seminiferous tubules as well as normal interstitial cells of Leydig with lack of congestion of interstitial blood vessels in the testis of rats treated with aluminum chloride with the concomitant treatment with propolis extract, these results agreed with Hasan et al. [66], who reported that the treatment with propolis antagonized aluminum chloride toxicity on liver and testis. The antioxidant property of propolis responsible for its protective effect in our results agreed with Chang et al. [67], Gómez-Caravaca et al. [68], and Mokhtar et al. [69] who reported that, propolis antagonized the harmful reproductive toxicity of aluminum chloride on testis. Propolis reduced the oxidative stress, apoptosis, and necrosis induced by aluminum chloride on the testes seen in the results approved by El Masry et al. [70], and Hasan et al. [66]. In the present study, co-administration of propolis practically prevented most of the toxic effects of aluminum chloride on the testis, these results agreed with Alyane et al. [71] who reported that pretreatment of rats with propolis extract reduced the peroxidative damage in the mitochondria as reducing both of mitochondrial malondialdehyde formation and the amplitude of mitochondrial swelling.
12. V. Conclusion
Our results demonstrated that the aluminum chloride induced toxic structural changes in the rat's testicular tissues, which could be improved by the protective effect of propolis supplement. These results validate the hypothesis that the testicular toxicity of aluminum chloride modulated by propolis supplementation.