1. Introduction
he skin is the largest organ in our body that provides a protective covering for the body besides its other physiological and defensive functions [1]. One of the most common skin pathologies is wound that defined as any break and disruption of the normal cellular and anatomical or functional continuity of the skin [2,3]. Wound is a multifactorial disorder and it could be caused by physical, chemical, electrical or microbial causes [2]. Returning of skin to normal structure and replacement of destroyed or damaged tissue by newly produced tissue is called wound healing. Wound healing is a physiological and complex process established by four continues and overlapping phases (hemostasis; inflammation; proliferation, and tissue remodeling) [4,5].
There are no lines of demarcation between the four previously mentioned stages that can be detected on the clinical or cellular level. The main goal of wound healing is to speed the recovery and restore the normal anatomical structure and function of the injured part with minimal scarring [6]. These steps involve multiple cellular and molecular events tightly controlled by numerous growthfactors, chemokines, and cytokines. In addition to the dynamic relationship between skin and microbiome (skin flora), that is another major contributor to the outcome of the wound healing process [7]. Signaling networks involving the interleukin (IL) family and growth factors are required to coordinate cell-cell and cell-extracellular matrix (ECM) interactions, that are essential to fully wound healing [8,9].
The program of healing process can be interrupted by numerous factors which are classified into local factors (oxygenation, woundinfection, foreign body, venous sufficiency, wounded part, depth, local tension, and pressure) and systemic factors (age, gender, sex hormones, stress, ischemia, diseases, obesity, medications, alcoholism, smoking, immunocompromised conditions, and nutrition) [10,11], Fig (1). Here in our paper, we highlight the variable applications of nanotechnology in veterinary medicine such as animal production, reproduction, diagnosis and treatment, nanovaccines, drug delivery, Nanosensors, and wound healing. Also, Chitosan nanoparticles were separately discussed.
Nanotechnology is a rapidly developing branch of science that studies the synthesis, structure, and dynamic of the atomic and molecular nanometer (maximum diameter of 100 nm) of nanoparticles [12]. Nanoparticles are synthesized by size reduction using either top-down methods such as milling, high-pressure homogenization and sonication or bottom-up processes such as reactive precipitation and solvent displacement [13]. The size and shape are key determinants to the biological efficiency of nanoparticles, by influencing active substance delivery (carrier circulation), penetrability (either directly through cell membranes or through phagocytosis), and cellular responses (receptor recognition) [14,15]. The convergence of nanotechnology with medicine has been adding new hope in the therapeutic and pharmaceutical fields through a new discipline known as nanomedicine. This is the way for the development of novel materials and for the treatment of several pathologies [16].
2. a) Nanotechnology in Veterinary Medicine
Nanotechnology application in veterinary medicine is still relatively innovative. Recently, the nanoapplications are already used in the veterinary medicine, animal health, production, breeding, reproduction, and nutrition [17], Fig (2).
3. Diagnosis and Treatment
In veterinary medicine, there is little definition of an animal's condition until clinical symptoms are detected. Infection can be substantially spread in the herd until disease is diagnosed. Nanotechnology may be effective method in critical clinical diagnoses because of this property [18].
Biochips can be used for diagnosis of animal diseases. A Biochip (or microarray) is a device typically made of hundreds or thousands of short artificial DNA strands that are accurately deposited on a silicon circuit. Biochips can also be used to trace the source of food and feeds to detect the presence of animal products from different species as a means to locate the source of pathogens a response to public health threats as avian flu and mad cow disease [19]. Using biochips, biological samples such as blood, tissue and semen can be instantaneously analysed and manipulated [20]. Nanoshells can be injected into the animal's to locate tumour cells. The nanoparticles release an attached drug at the tumor site to destroy the cancer cells. [26]. Also it has a role in reduction of aflatoxin as in use of nanosilver which used in treatment of experimental aflatoxicosis in broiler chickens. In other study revealed that supplement of nanosilver to diet containing aflatoxin could diminish the inhibitory effects of aflatoxin [27]. Also, gold nanoparticles and gold-based test strip have been used for the rapid detection of infectious bursal disease (IBD)virus antibodies in chickens[28], foot-andmouth disease (FMD) virus [29], pathogenic bacteria [30], bluetongue virus [31], specific bacterial contaminants in chicken, such as S. typhimurium and S. enteritidis [32], antibacterial effect against Bacillus Calmette-Guérin (BCG) and Escherichia coli [33], haptoglobin in mastitic milk of bovine [34], diagnosis of viral infections in pigs [35], rapid detection of Mycoplasma suis in porcine plasma [36], and bacterial toxins [37]. Mastitis is common multifactorial disease that affects high-yielding animals primarily cows and is usually caused by Staphylococcus, Streptococcus, and E. coli, which leads to economic consequences for reducing the yield of milk [38,39]. Zinc oxide nanoparticles (ZnO-NPs) have been found to be effective against bacterial biofilms inside the udder tissue such as S. aureus and E. coli [40,41]. ZnO-NPs is more sensitive to detect cancer biomarkers and used in diagnostic and therapeutic purposes in common neoplastic conditions of animals such as lymphoma, cutaneous cancer, transmissible veneral tumor, and equine sarcoids [42][43][44] .
In experimental studies applied by [45] on recent and infected wound in veterinary field by application of chemical and green synthesized ZnO NPs, they concluded that NPs having excellent antimicrobial healing effect with minimal scarring especially in green synthesized ZnO NPs. Also, Nanoparticles could be used in mass treatments of ectoparasites facilitating the drug-parasite interaction. Nanoparticles are used for improvement of the effectiveness of phytotherapeutic products such as theantiparasitic silver nanoparticles using Cissusquadrangularis against Hippoboscaaculata and Rhipicephalus (Boophilus) microplus [46]. The nanoshells facilitate the target of the parasite improving the treatment of parasites and it can be useful in the study of host-parasite as biosensors. Nanodevices can allow the diagnostic of parasite in herds allowing veterinarians to suggest a correct health management [47].
4. Nanovaccines
Nanotechnology provides different forms of veterinary nanovaccines such as: recombinant Bacillus anthracis spore-based vaccine, influenza virus vaccine, Heliobacter pylori vaccine, Tetanus toxoid, Bordetella pertussis vaccine, Rota virus capsid vaccine and Bovine parainfluenza type 3 vaccine. The mucosal immunity is developed following intranasal administration of influenza virus vaccine and both IgG and IgA immune responseare developed following the oral administration Bovine parainfluenza type 3 vaccine.
Also chitosan nanoparticles (CS NPs)were prepared with Leishmania superoxide dismutase and subcutaneously injected as recombinant Leishmania SOD vaccine. Also, intranasal (pneumococcal antigen A vaccine and Streptococci equi vaccine)or pulmonary (TB vaccine) vaccines were nano-based. Also, gold nanoparticle-based vaccine were developed against FMD, Newcastle disease, influenza virus, or herpes simplex virus 2, and also vaccines of African horse sickness virus (AHSV) [48][49][50].
5. Drug Delivery
The nanoparticles play an important role in drug delivery as they can carry drug and genes to the target site. The drug delivery systems have important effects on absorption, distribution, and excretion rate of drugs or other substances in the body [6]. Liquid vitamins that were prepared by nanotechnology are available for poultry feed and their nanosized are designed to pass through the gastrointestinal tract and deliver the vitamins or other nutrients directly in the bloodstream and therefore increase their bioavailability. NPs mask undesired tastes and improve the nutrient dispersibility and durability of the feed. In addition, they decrease the need to use preservatives [51]. In animals, smart drug delivery systems allow antibiotic use in amounts less than possible. Also, smart delivery systems can perform processes such as its ability to decide the regulation in the process of delivery of drug or its ability to make the drug addition according to the need, and the perception of chemicals in the site. Smart delivery system decrease the antibiotic usage and the treatment cost in farms [52].
6. Nanosensors
Nanosensors are miniature devices that can be used for diagnosis of biological material or tissue samples [53]. It has been used in several biomedical applications in veterinary medicine including animal health for diagnosis of some microbial pathogensdiseases [54,55] and for animal production, especially the delivery of controlled quantities of drugs into breeding animal beverages. Nanosensors used for prevention of bovine tuberculosis [54] and early diagnosis of subclinical Ketosis (SCK) that is essential for management of dairy cattle health [56]. Detection of Mycobacterium bovis using nanosensors in infected cattle via breath allowed for real-time cattle monitoring [57]. In addition, the sensors can be used to measure body temperature, antibiotic detection, detect their sweat constituents, observation of behavior, movement, and stress via the implantation on animals [55].
7. Animal production and Food sector
By experimental studies applied by [58] on New Zealand White rabbits fed on nanoparticles; they showed increased weight gain at the fattening period. Similar beneficial production effects have been reported by [59] and [60] in chicken experiments. As the excellent results to an additive which improves the sanitary status of animals. So, the beneficial effect of nanoparticles used in animal production was most frequently observed in such parameters as weight gain, average daily gain, or improvement of the feed conversion rate (FCR) value [61]. As well, the nanomaterials that have been digested and shed in the feces can be used for fertilization in pastoral soil.
Nowadays, the nanomaterials are used for meat production as ingredients or additives in the food. They enhance the taste and reduce the use of fat, salt, sugar, and preservatives. Reducing the salt level is especially important and presents a great challenge for meat industry because in spite of advantages, use of salt consequently increased risk of cardiovascular disease since it is linked to hypertension [62].
8. Animal reproduction
Management of breeding is an expensive and time-consuming problem for dairy and swine farmers [19]. There are various applications of nanotechnology in the field of animal reproduction which optimize the general reproductive performance at its different stages starting from the diagnosis and treatment of reproductive disorders and detection of estrus till managing reproductive problems such as retained placenta [63]. Also, the NPs can also be used for sustained release of reproductive hormones [64].
Currently, Nanosensors also used to diagnose genital tract infectious diseases, metabolic and hormonal disorders, and even for the detection of estrus [65,66]. Similarly, a nanotube implanted under the skin, it is bind and detects the estradiol antibody at the time of oestrus by near infrared fluorescence and the change in level of estradiol in the blood can be measured [19].
9. b) Chitosan Nanoparticles (CS NPs)
Chitosan Nanoparticles (CS NPs)is an excellent example of nanomaterials which included in all stages of wound healing [67]. CS NPs showed an increased activity than its parent chitosan [68] due to its increased surface to volume ratio [69]. Chitosan is a natural and biodegradable polysaccharide composed of ?1?4 linked N-acetylglucosamine and derived from the alkaline deacetylation of chitin which is the principal component of the cell walls of some fungi and the exoskeleton of insects, crustaceans [70,71], mollusks [72], yeast or green algae [73].
10. Synthesis
CS NPs consists of three functional groups, including an amino group and primary and secondary hydroxyl groups. CS NPs can be cross-linked with glutaraldehyde, glyoxal, and terephthaldehyde, producing hydrogels that can be used in different applications such as organ transplants, restoring organ function, gene delivery, and targeted therapy [74,75]. so, chitin and chitosan are manufactured commercially in large scale [76]. In its crystalline form, chitosan is normally insoluble in an aqueous solution above pH 7; however, in diluted acids (pH 6.0), the protonated free amino groups on glucosamine facilitate solubility of the molecule. Chitosan preparations of various molecular weights (50-2,000 kDa), degrees of deacetylation (30-95%), and further molecular derivatization patterns allow extensive adjustment of mechanical and biological properties [77].
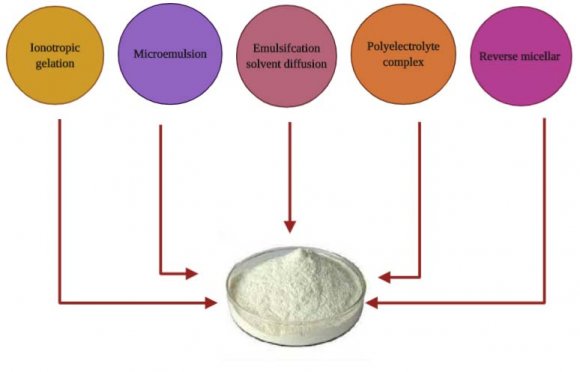
CS NPs can be synthesized using either "bottom-up" or "top-down" approaches and/or a combination of both procedures [78]. CS NPs are synthesized through numerous "bottom-up" approaches such as polymerization of CS with methacrylic acid (PMAA) to make CH-PMAA NPs, or through a reverse micelle medium or microemulsion methods [79]. Topdown approaches such as milling, high-pressure homogenization, and ultra-sonication are also applied for the synthesis of these nanomaterials [80]. Many methods have been employed for the syntheses of CS NPs, but five methods are presently available. They are ionotropic gelation, microemulsion, emulsifcation solvent diffusion, polyelectrolyte complex, and reverse micellarmethod [81], Fig (3). Out of this, the most widely used methods are ionotropic gelation and polyelectrolyte complex. These methods are simple and do not apply high shear force or use organic solvents [82].
11. Biomedical applications
Chitosan nanoparticles have great commercial value due to its high nitrogen content and other physiochemical properties like biocompatibility, adsorptive ability [69], biodegradable [72], anticholesterolemic, fungistatic, noncarcinogenic, remarkable affinity to proteins, stimulation of healing, tissue engineering scaffolds, and drug delivery [83][84][85]. In addition to, hemostatic activity, anti-inflectional activity, property to accelerate wound healing [86], allergenicity, and bioactivity make it a very attractive substance for diverse applications as a biomaterial in the biomedical, food, and chemical industries [87,88]. Other chitosan properties are low toxicity, availability, low cost, permeability to oxygen, and ease of chemical modification [70,89,90]. The biodegradation rate of chitosan samples is the highest at a temperature of 36°C and is characterized by a relatively prolonged induction period, which is shorter at higher temperatures. The smaller the molecular weight of chitosan, the shorter the induction period [91].
12. Drug Delivery
Separated reports are available on the use of combinations of antibiotics and chitosan and its derivatives as antimicrobials. Decker et al. proposed a synergistic chlorhexidine/chitosan combination for improved anti-plaque strategies [92]. Tobramycin is one of the antibiotics which is reported to show synergistic action with chitosan in planktonic culture of Pseudomonas aeruginosa [93]. Bioadhesive and antimicrobial properties of chitosan and its derivatives are effective in antimicrobial drug delivery control, release of Chlorhexidine and Nystat in oral preparations [94,95], release of ampicillin [96], drug delivery system for Ofloxacin [97] in ophthalmic preparations, and gastrointestinal-release preparations for Tobramycin sulfate [98].
13. Antimicrobial and antifungal activities
CS NPs are present in many forms such as beads, powders, gels, sponges, tubes, fibers, and films [99][100][101], hydrogel [102], and biofilm [103]. These forms facilitate the antibacterial and wound-healing activity of CS NPS. Chitosan nanoparticles were shown to have significant bactericidal effects on different types of bacteria and no cytotoxic effect on mouse fibroblast cells [104]. Their antimicrobial effects against a broad range of gram positive and gram-negative bacteria as well as fungi were reported [105][106][107]. Archana, D., et al., reported in an experiment on a wound in rats the antimicrobial and wound-healing activity of chitosan [67]. Although several studies have shown that chitosan exhibited higher antibacterial activity against grampositive bacteria than gram-negative bacteria, while some other studies have shown that gram-negative bacteria as more susceptible than gram-positive [108,109], many works have demonstrated that there is no significant difference between antibacterial activity and bacterial species [110].
Studies have reported the antibacterial activity of CHNP against Escherichia coli, Staphylococcusaureus, Streptococcusmutans, Salmonellatyphimurium, Salmonellacholeraesuis, and Pseudomonasaeruginosa [107,111]. Antifungal activity of CHNP has also been reported against Aspergillusniger, Fusariumsolani, Rhizoctoniasolani, Collectotrichumgloeosporioides, Candidaalbicans [68,112]. Chitosan's antibacterial efficacy also depends on multiple, factors both intrinsic and extrinsic. Intrinsic parameters comprise the molecular weight and degree of deacetylation of parent chitosan, nanoparticlesize, and concentration. The extrinsic factors involve pH, temperature, reactive time and so on [113].
The CS NPS size is an important factor which affects its antimicrobial activity, as the decrease in size at lower concentrations is due to decreased viscosity causing better solubility of chitosan in acetic acid solution and resulting in better action of chitosan [114].
However, deacetylation degree, positive charge content, and other factors including molecular weight can affect the antibacterial activities of chitosan [115], as the molecular weight has a stronger effect on the antimicrobial activity compared to the degree of acetylation [108,116,117]. It has been reported that decreasing the molecular weight of chitosan may increase its binding affinity to the membrane due to improved mobility, attraction, and ionic interaction [118], though a proper antibacterial activity can be obtained only when the molecular weight is larger than 10 kDa [119]. In acid, the antimicrobial activity was shown to increase with increasing molecular weight [116], while the antimicrobial activity changed at pH 7.0 [115].
The most prevalent proposed antibacterial activity of chitosan is by binding to the negatively charged bacterial cell wall causing disruption of the cell thus altering the membrane permeability [120,121], followed by attachment to DNA causing inhibition of DNA replication and subsequently cell death [121]. Another possible mechanism is that chitosan acts as a chelating agent that selectively binds to trace metal elements causing toxin production and inhibiting microbial growth [122]. For instance, chitosan composites have shown antiviral activity against H1N1 influenza A virus [123] and Human cytomegalovirus (HMCV) strain AD-169 [123,124].
14. Wound healing
The application of nanotechnology in the field of wound healing gave way to the manufacture and innovation of many bandages, which in turn speed up healing with the least possibility of scarring. [16]. From our point of view, the good selection of suitable nanomaterials for different wound types can play the main role in wound healing and tissue remodeling. The nanomaterials used in wound healing are classified into two categories: (1) nanomaterials that exhibit intrinsic properties beneficial for wound treatment and (2) nanomaterials included in drug delivery [125,126]. An An experimental study performed by Chandika, P., et al [128] documented that chitosan increased wound healing rates in mice. N-acetyl glucosamine (NAG) present in chitin and chitosan is a major component of dermal tissue which is essential for repair of scar tissues [129]. Chitosan's positive surface charge enables it to effectively support cell growth [130] and promotes surface-induced thrombosis and blood coagulation. Free amino groups which are present on the chitosan membrane surface may form polyelectrolyte complexes with acidic groups of the cellular elements of blood [86]. Chitosan promotes the dermal regeneration during skin reconstruction by regulating the deposition and accelerating the arrangement of thin collagen fibers. It also modulates fibroblast activation, sustaining the regeneration of the extracellular matrix while stimulating cell proliferation and migration (i.e. the formation of granulation tissue) and enhancing wound healing [131].
Natural products-derived biomaterials can provide materials with greater complexity and composition, and in order to mimic the extracellular matrix (ECM) conditions of the wound and to provide a scaffold for the fibroblasts for collagen deposition, ECMbased therapies have gained popularity [132] .
Studies suggest that chitin and chitosan promote the migration of polymorphonuclear and mononuclear cells and accelerate the reformation of connective tissue and angiogenesis (necessary for the arrival of repair cells such as fibroblast) [133]. In recent studies, chitosan has been used to deliver bioactive molecules, basic fibroblast growth factor [86] and human epidermal growth factor [134] were encapsulated in this biomaterial; electrospun nonwoven nanofibrous hybrid mats based on chitosan and poly[(L-lactide)-co-(D,L-lactide)] were produced; [135] chitosan dressing incorporating a procoagulant (polyphosphate) and an antimicrobial (silver); [85] and chitosan acetate bandages were used as a topical antimicrobial dressing for infected burns [83].
15. II.
16. Conclusion
Nanotechnology is a rapidly developing branch of science that could be applied in various aspects with different actions. Although advent of nanotechnology in veterinary medicine isn't similar as in human medical settings, but this field has been recently attracted veterinary researchers. We conclude that the field of nanotechnology in veterinary medicine is a promising field in different disciplines; surgery, pharmacology, vaccinology ... etc. 17. Manuja