1. Introduction
iposarcomas are rare malignant tumours of adipocytic differentiation and are classified under the soft tissue sarcoma subtype histologically. Retroperitoneal liposarcoma (RPL) has an incidence of 1 per 2.5 million individuals, and the retroperitoneum is the second most common site for a liposarcoma, following the lower limb as the most common site. The retroperitoneum is a space that can easily expand;therefore tumours grow in this space without any symptoms until they are very large.
This case study reports a patient diagnosed with a retroperitoneal liposarcoma treated with primary radical surgery and the patient is currently being followed-up in our unit with close monitoring and Computed Tomographic (CT) imaging. The patient consented to the reporting of this case.
2. II.
3. Case Report
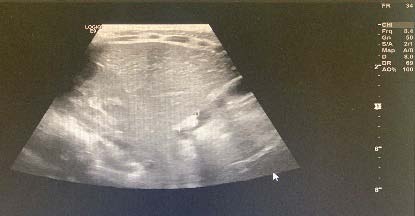
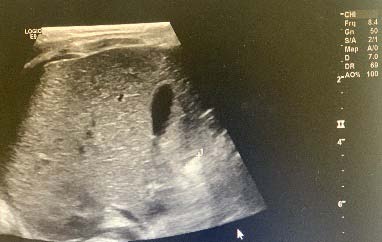
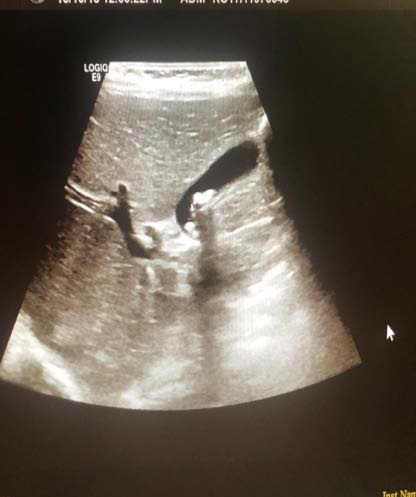
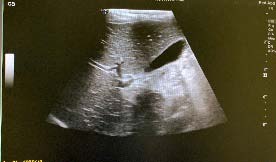
A 70-year-old female patient was referred to our unit for assessment of anasymptomatic solid left sided adnexal mass seen on CT scan during evaluation for aurological complaint.
At the time of the referral the mass measured 5.5 x 4.9 x .7.2 cm with a macroscopic fat and a large soft tissue component on CT-scan assessment. Tumour markers were all essentially normal, CA 125 of 15 U/mL, CA 19.9 of U/mL, CEA of 2.8 ug/L and the AFP 13.4 k/U, which is a slightly raised level. During this assessment, an excisional procedure was offered to the patient, but due to the Covid 19 pandemic the patient opted to wait with a surgical procedure and follow-up at a later stage.
The patient presented 9 months later for reassessment. During this time the mass had increased significantly in size and had become symptomatic and palpable in the left lower quadrant of her abdomen. On repeat CT-scan the mass appeared to have significantly increased in size, measuring 12x 8 x 14 cm. The mass still had a predominantly solid appearance and was strongly associated to the left ovary and likely ovarian in origin. Ovarian cancer markers were repeated and as above stayed within normal limits. Due to the appearance of the lesion and the very rapid growth, a non-benign lesion, possibly of ovarian origin was suspected and an explorative/staging laparotomy was offered to the patient. The patient consented to the procedure. A mid-line laparotomy was performed, and a large retroperitoneal mass was found. This mass was adherent to the left ovary, left ureter, left psoas muscle and external iliac vessels as well as the sigmoid colon. With careful anatomical dissection the mass was resected, and a full staging laparotomy and lymph node sampling of the left pelvic and para-aortic lymphnodes was performed. The specimen comprised of a 527g encapsulated, lobulated portion of tissue, with a histological diagnosis of a well differentiated liposarcoma, sclerosing variant. No areas of dedifferentiated liposarcoma were noted and the margins were clear of tumour. All resected lymph nodes were negative for metastatic disease. The patient had an uneventful post-operative recovery.
Although the retroperitoneal liposarcoma appears to have been removed completely in this patient, local recurrence is likely. Since there is currently no evidence that radiotherapy or chemotherapy improves survival rates or recurrence rates of the disease in the immediate adjuvant setting, the patient did not receive any adjuvant therapy and is being monitored closely with clinical examinations and 3 monthly CT-scans to evaluate for any local or distant recurrent disease.
4. III.
5. Discussion a) Surgical and Anatomical features
The retroperitoneum and the preperitoneum forms the extraperitoneal space, which is the portion of the pelvis and abdomen which does not lie within the peritoneal space. The retroperitoneal space is an almost virtual and expandable space, defined anteriorly by the peritoneal extensions anchoring the transverse colon, the small bowel as well as the 1 1 ascending and descending colon, part of the duodenum, part of the pancreas and part of the liver 2 3 L . The retroperitoneum contains the kidneys, the adrenal glands, the pancreas, part of the duodenum, ascending and descending colon, the abdominal aorta and vena cava (dividing into the common iliac, external and internal iliac arteries and veins respectively), the abdominal lymphnodes groups and tracts, six major nerves and the autonomic lumbar nerve chains and the connective tissue of fasciae, with the White line of Toldt as the fusion between the mesocolon and the posterior retroperitoneum.
Treatment of most conditions involving the retroperitoneum requires surgical intervention. A complete and thorough understanding of the anatomy of the structures involving the retroperitoneum is prudent in avoiding inadvertent damage to underlying structures. Complications may arise from inadvertent damage to structures located within the retroperitoneum during surgical manipulation or instrumentation 4 b) Histopathological features .
Retroperitoneal liposarcomas usually consist of a large, well-circumscribed, lobulated mass. Variable consistencies are present, from yellow to firm grey to gelatinous areas, depending on the proportion of fat, fibrous and myxoid components. Larger retroperitoneal tumours appear more heterogeneous, often containing foci of fat necrosis and punctate haemorrhages 5 .
6. Figure 1: Macroscopic appearance of the excised retroperitoneal liposarcoma
As in many tumours, the histological classification of liposarcoma has evolved over the past several decades, mostly owing to the advances in our understanding of molecular genetics.
The most recent World Health Organisation (WHO) classification system (2020) recognized five different types of major liposarcoma subtypes, differentiated by distinctive morphologies and unique genetic findings 5 ? Atypical lipomatous tumour (ALT)/ well differentiated liposarcoma (includes adipocytic [or lipoma like], sclerosing and inflammatory variants) :
? Dedifferentiated liposarcoma ? Myxoid liposarcoma ? Pleomorphic liposarcoma ? Myxoid pleomorphic liposarcomaIn atypical lipomatous tumour/well differentiated liposarcoma adipocytic variant consists out of cells that vary substantially in size as well as cells that have Dedifferentiated liposarcoma can arise as a synchronous lesion in 90% of cases and as metachronous lesion in 10% of cases 6 . These tumours exhibit a wide morphological spectrum and histologically show areas of high grade, poorly differentiated sarcoma resembling high-grade myxofibrosarcoma, fibrosarcoma, malignant solitary fibrous tumour or pleomorphic sarcoma not otherwise specified. Dedifferentiated liposarcomas can be of variable histological grade 5 . Dedifferentiated liposarcomas is an aggressive disease, arising most commonly in the retroperitoneum and is associated with high rates of local and metastatic recurrence and disease specific mortality 7 Use of the term atypical lipomatous tumour is determined by tumour location and resectability. In locations such as the retroperitoneum, it is usually impossible to obtain a wide tumour free surgical margin of more than 2cm, thus local recurrence is common and leads to mortality, seen in the absence of dedifferentiation or metastases The introduction of computed tomography (CT), magnetic resonance imaging (MRI), and ultrasonography have greatly enhanced our capability to make the diagnosis of abdominopelvic neoplasms and determine and direct treatment, as well as observe the progress of the neoplasm and effect of treatment over time. Identification of a retroperitoneal mass at imaging is a challenging task for radiologists, however the presence of fat within a retroperitoneal lesion is helpful in refining the differential diagnosis 9 ? On ultrasound appearance it is hyperechoic and may demonstrate posterior acoustic shadowing . It is easy to recognize fat within a lesion due to its characteristic imaging appearance:
? Computed Tomographic (CT) imaging derives contrast parameters predominantly from the physical properties of tissue -in combination with high resolution spiral CT, this provides accurate attenuation measurement, with -10 to -100 HU9corresponding to fat 10 ? Magnetic resonance imaging (MRI) has lower spatial resolution than CT imaging, but better soft tissue contrast and greater sensitivity in detection of microscopic fat. The two MRI techniques for the identification of fat within a structure are fat saturation and chemical shift imaging It can be difficult to localize large abdominal masses to an anatomical space and to accurately identify the organ of origin thus to determine whether the lesion arises from a retroperitoneal organ, or from the soft tissue 10 . If the mass is surrounded by the parenchyma of the organ, it undoubtedly arises from the organ, however if the mass originates at the margin of the organ, it is more difficult the determine the origin. The interface between the mass with the adjacent organ can provide insight as to whether it displaces the organ or arises from the organ (Table 1 Organ of origin is obliterated by the tumour Prominent feeding artery sign
Large feeding arteries of a hypervascular to the of origin A CT image of a lipoma will reveal a welldefined homogenous mass with fat attenuation. Areas of soft-tissue attenuation may be seen within the tumour and may represent fat necrosis, septa or normal adjacent structures. If a predominantly solid soft-tissue component or adjacent organ invasion is present, a liposarcoma should be suspected. The imaging characteristics of liposarcomas differs, depending on the histological subtype.
Well-differentiated liposarcomas appear as well defined predominantly fat-containing lesions with minimal soft tissue attenuation and commonly contain septa -the appearance may be indistinguishable from a lipoma 9 and therefore aretroperitoneal purely fatty lesion should be considered a liposarcoma rather than a lipoma until proven otherwise with histological confirmation 10 11 . Dedifferentiated liposarcomas appear remarkably similar to well-differentiated liposarcomas of CT imaging, and dedifferentiation is suggested by focal nodular non-lipomatous regions larger than 1 cm 12 Surgery is the mainstay of treatment for nonmetastatic retroperitoneal sarcoma 8 . If possible, macroscopically complete resection of tumour should be aimed for and this can lead to radical surgery requiring en-bloc removal of adjacent structures. If the pre-treatment diagnosis can be made with certainty, based on radiologic and clinical findings and complete resection is deemed possible, pre-treatment biopsy is not advised and has no value 13 . If radiologic investigations suggest a pathology that does not require primary surgery (e.g. lymphoma, Ewing Sarcoma, GIST) or the incomplete resection is expected, biopsy will be necessary to plan alternative treatment. Image guided core biopsy is advised and preferred over open or laparoscopic approaches, which may be associated with tumour spillage and compromise future surgical strategy by altering tissue planes 14 The removal of the entire tumour with a margin of normal tissue is usually not possible in large retroperitoneal liposarcomas due to the presence of adjacent large vessels, nerves and bony structures, leading to local recurrence in the abdomen, which constitutes the cause of death in three out of four patients . 15 ii. Radiation Therapy . High grade, dedifferentiated tumours are at a higher risk to recur and spread systemically, so even if extensive surgery with adequate margins is achieved, the prognosis remains dismal, querying the fact whether a patient should be exposed to the morbidity of extensive surgery if the mortality in dedifferentiated liposarcoma remains high irrespective of treatment.
Currently there is no convincing evidence for the role of radiotherapy (RT) in the adjuvant setting for the management of RLS. Several authors have analysed the data from the surveillance, epidemiology, and end result (SEER) data base in order to define the role of adjuvant RT in RLS and as a general finding it did not improve survival or did so in a subgroup of patients with stage I disease only. To date, no randomised trials have been completed or published comparing surgery alone with combined surgery and RT. Pre-operative RT in certain settings is showing some promise, however further studies and data would be needed. iii. Systemic therapy Chemotherapy has an established role in the palliative management of advanced or metastatic soft tissue sarcoma 8 .
Active agents include the anthracyclines (doxorubicin and epirubicin) and the alkylating agent ifosfomide 16 . In patients with resistant disease, gemcitabine, docetaxel, trabectedin and pazopanib were established as effective second-or third-line options in the recent years 17 The response of liposarcoma to chemotherapy differs according to histological subtype and grade . 8 . Well-differentiated and dedifferentiated liposarcoma respond poorly to systemic therapy, therefore novel molecular targets will have to be identified to explorenew possibilities for treatment. MDM2 and CDK4 targeted therapy as well as the tyrosine kinase inhibitor Sunitinib is currently showing promise in treatment of RLS 8 .
7. IV.
8. Conclusion
Retroperitoneal liposarcoma is a rare malignancy and challenging to diagnose, treat and monitor for recurrence. The presence of ahigh-grade dedifferentiated component does make the disease more aggressive, but even well differentiated tumours can be difficult to manage and recurrences can be widespread and higher grade than the primary tumour.
Even though the mainstay of treatment is surgery, multi-disciplinary discussion is paramount, especially in view of anatomically irresectable sights and potential future benefits of non-surgical therapies. As with any rare malignancy national and international collaboration is encouraged to learn from each other and improve management of retroperitoneal liposarcoma.

| D D D D ) F |
| ( |