1. Introduction
M is a metabolic disorder characterized by high levels of free glucose in the blood. Diabetes is a disorder in the human body that causes blood glucose (sugar) levels to rise above normal due to insulin secretion or insulin resistance. This is also called hyperglycaemia (1). DM is an incurable disease that has a detrimental effect on many metabolic pathways and contributes to the pathophysiology of diabetic complications (2,3). Oxidative stress is a very important factor in the development of diabetes (8). Oxidative stress is known to be associated with lifestyle-related diseases including atherosclerosis, high blood pressure, and diabetes. Free radical forms are important for body parts in biological homeostasis (11,12), but where their production is excessive and greater than the body's antioxidant capacity, then the effects of oxidative stress (12). Oxidative stress is a major factor in the development of diabetes and insulin resistance (12)(13)(14), triggering pathophysiologic pathways and initiating a burst of malignant pathways leading to insulin resistance and DM (8,15). In this review, we discussed the potential roles of oxidative stress in building insulin resistance and DM.
2. II. Classification of Diabetes Mellitus
There are different types of DM, but the most common subtypes are 1 DM (T1DM) and 2 DM type (T2DM). T1DM occurs due to beta-cell dysfunction, reduced insulin secretion, and low levels of circulating insulin. T2DM is the most common type of DM that accounts for approximately 90-95% of patients with diabetes and is mainly associated with insufficient response to insulin (reduced insulin sensitivity) and insulin resistance in borderline tissues (16). With type 2 diabetes, the human body does not use insulin properly. This is called insulin resistance. Initially, pancreas produces extra insulin to make it. Over time the pancreas may not be able to cope and it may not produce enough insulin to maintain normal blood sugar levels. Type 2 can be controlled by improving lifestyle, oral hypoglycaemic therapy, and insulin. Pregnancy diabetes is another topic that occurs in women during pregnancy when the body is less sensitive to insulin. Pregnancy diabetes does not occur in all women and usually develops after childbirth (17). Other types of DM are adolescent diabetes which is diabetes mellitus, preexisting diabetes in adults, and secondary diabetes from other diseases such as pancreatitis or secondary to the use of drugs such as corticosteroids (18,19).
3. III. Oxidative Stress and Antioxidant Process
Various normal cells produce free radicals such as aerobic respiratory products and other metabolic processes (7) including reactive oxygen species (ROS). ROS is a highly active oxidant and can have adverse effects on cellular lipids, proteins, and DNA or reactive oxygen species (ROS) produced by organisms due to normal cell metabolism and environmental factors, such as air pollution or cigarette smoke. ROS are highly active molecules and can damage cell formation such as carbohydrates, nucleic acids, lipids, and proteins and alter their functions. Cells usually contain enzymes and coenzymes that act as antioxidants. This helps to reduce ROS and prevents it from causing damage (6). Oxidative stress is defined as an imbalance between the chemical processes responsible for the production of active oxygen (ROS) and those responsible for the removal of ROS (20). There are many enzymes in the cell that have internal mechanisms such as superoxide dismutase (SOD), catalase (CAT), and glutathione (GLT), which protect cells from free radicals (25). Some heavy metal products have free properties such as iron (ferric) and copper (26) that can mix proteins, lipids, and nucleic acids and produce toxic products that lead to tissue dysfunction (27,28). They alter the structure of biologic molecules and break them down (28). DNA fragmentation is a well-known result of oxidative stress, which affects genetic expression and cell survival (23). Malondialdehyde (MDA), total cholesterol, and active hydroperoxides (ROOH) are oxidative stress biomarkers that occur in diabetic patients. (30). Oxidative stress plays important roles in diabetic complications through lipid peroxidation, DNA damage, and mitochondrial dysfunction (13,23,31,32). Its involvement has been shown in other illnesses and age-related problems such as cardiovascular disease, chronic obstructive pulmonary disease, chronic kidney disease, neurological diseases, and cancer and more. Including high free varieties (33). Many scientists believe that the theory of oxidative evolution is a major cause of aging and related problems (33). Problems caused by oxidative stress and insulin resistance are prevented with the help of redox biology (34).
4. IV. Normal Insulin Signaling Pathways and Insulin Resistance
Insulin is usually a 51 amino acid dipeptide containing a series A and B series linked to 2 disulfide bonds found in cysteine residues. The chain contains 21 amino acids and the B chain contains 30 amino acids. Insulin is encoded by a short arm of chromosome 11 in pancreatic ?-cells containing signal peptide, chain B, connective peptide (and A) and A chain (4,5). In proinsulin, C-peptide binding is enclosed at each end by the dibasic residues (Arg-Arg and Lys-Arg) that link the N-terminus of the A series and the C-terminus of the B chain (124,125). Proinsulin made from Golgi substances is converted to insulin by removing dibasic residues by trypsin-like endyprotease enzymes such as insulin and C-peptide (5). Insulin resistance is a key factor in T2DM where cells are unable to respond to insulin effectively (8,35) There are different enzymes and mediators, which facilitate the entry of glucose into adipocytes, muscles, and myocardial cells via GLUT-4 (glucose transporter-4) transporters [8,34]. The feature is triggered by binding insulin to ? chain of insulin receptors (IRs), which are members of transmembrane tyrosine kinases that are made up of ? and-chains and activated by insulin and IGF-(insulin-like growth -) 1 and IGF-2 (36). As a result, binding induces structural changes in the chain by autophosphorylation in tyrosine residues through different adapter proteins, namely, insulin receptor substrates (IRSs), Shc proteins (SHCtransforming), and APS protein (protein adapter) (37,38)). These processes provide a suitable site for binding IRS-1 (insulin receptor substrate-1) (38). Many types of insulin-dependent kinases such as extracellular protein kinase C, S6K1 (ribosomal protein S6 kinase beta-1), serine / -threonine-protein kinase 2, protein kinase B etc and other types of kinases such as AMPactivated protein kinase and glycogen synthase kinase 3 can activate and activate phosphorylate IRS (38,39). The activated IRS-1 binds to PI3K (phosphoinositide 3kinase) and activates it, which, in turn, promotes the conversion of PIP2 (phosphatidylinositol 4,5bisphosphate) to PIP3 (phosphatidylinositol 3,4,5trisphosphate) (40). PIP3 itself is a potent activator of Akt, which also contributes to cell-induced glucose uptake through the production of GLUT-4 and inhibits glycogen synthase kinase leading to increased glycogen secretion (40,41). Any disruption in the abovementioned steps may cause insulin resistance and DM (34).
5. V. Relationship between Oxidative Stress and Insulin Resistance
Oxidative stress increases the risk of insulin resistance and DM (26,34). It should be noted that oxidative stress caused by DM has more complex interactions (42,43). The following are potential molecular mechanisms by which free radicals disrupt the normal glucose Homeostasis contributes to the formation of DM.
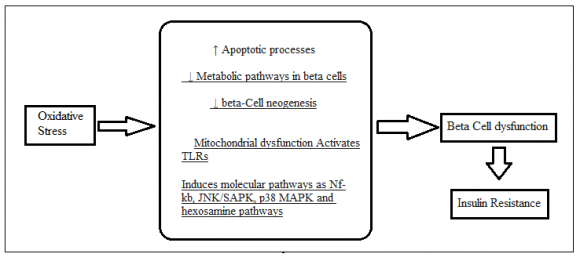
6. a) ?-Cell Dysfunction/Insulin Production and Secretion
The function of insulin is to maintain blood glucose levels by promoting glucose uptake into insulintargeted tissues. Glucose is a key regulator of insulin secretion by pancreatic Beta cells, which triggers a burst of events called beta-stimulating cells -the ability to sense circulating blood sugar levels and release the right amount of insulin to keep blood glucose at a normal level (126)(127)(128)(129).
Normal glucose homeostasis is made up of healthy and active beta cells and DM is associated with various levels of beta-cell dysfunction (44,45). DM occurs as a result of loss of beta-cell function and function (46). In these cases, insulin secretion produced by glucose from beta cells is reduced and decreased; therefore, glucose levels have risen above normal levels (47). In this process, insulin secretion occurs, which is interrupted by a decrease in the energy of the sugar to promote insulin secretion leading to severe unstable insulin release and followed by beta-cell failure (46). Beta-cell dysfunction occurs due to pathogenic mechanisms and oxidative stress (46,48). Free radicals in pancreatic beta cells arise due to enzyme activity such as mitochondrial respiratory tract (MRC) and NADPH (nicotinamide adenine dinucleotide phosphate) oxidase or NOX enzyme (15,(49)(50)(51). Superoxide anion is a major free radical pathway produced by MRC and NOX enzyme in beta cells (52). Beta cells are affected by free radicals produced by phagocytic and immune cells (53). Chronic hyperglycemia induces free radical production in the islands by increasing cytosolic calcium and protein kinase activation pathways (50,54). Beta cells have a low dose of antioxidant protection system, so that oxidative stress on beta cells increases in DM and plays an important role in the loss of their function in both T1DM and T2DM (45,48,55). Oxidative stress disrupts beta-cell function through a number of molecular mechanisms (48,(55)(56)(57)(58)(59)(60)(61)(62). Significantly reduces insulin production, disrupts the insertion of proinsulin vesicles into plasma membranes, and reduces their exocytosis in response to glucose distribution (48,55,56). It can also cause apoptotic processes in pancreatic cells that lead to death and loss of beta cells (48, 55, 56,). Therefore, depressive beta cell dysfunction caused by stress is a major potential target for experimental intervention in patients with DM. We suggest that pharmacologic agents protecting the islands from oxidative damage may provide Target therapeutics to promote beta-cell function that leads to improved glucose homeostasis. GLUT4 is an insulin-regulated glucose Transporter found in adipose tissue and bound muscles such as skeletal and heart muscle and, therefore, to maintain insulin sensitivity in these tissues a normal body profile of GLUT-4 expression and / or localization is required (15,63). The reducing factor of the GLUT-4 antagonist has an effect on insulin sensitivity (63,64) as a decrease in glucose entering target cells translates into lower insulin sensitivity in these tissues (65). According to Clinical Studies GLUT-4 expression and / or localization decreased insulin-resistant and T2DMresistant patients (64,66,67). This patho physiologic condition is exacerbated by oxidative stress (68,69). Oxidative stress can reduce the content of GLUT-4 by impairing gene expression by damaging the binding of the nuclear material to the GLUT-4-induced insulin receptor in 3T3-L1 adipocytes (70). 3T3-L1 adipocytes develop oxidative stress in these cells and receive Glut-4 expression from these tissues. They found that hydrogen peroxide produced by significant oxidative stress is regulated by GLUT-4 to 3T3-L1 adipocytes and, consequently, reduces cellular glucose (70). Other studies suggest that oxidative stress reduced GLUT-4 transport to cell membranes. They induced mitochondrial oxidative stress using mitochondriatargeted paraquat to adipocytes and found that oxidative stress significantly reduced GLUT-4 transport and thus induced insulin resistance in these tissues (71). Long-term oxidative stress can suppress transcription factors involved in GLUT-4 expression such as peroxisome proliferator-activated receptor gamma, CCAAT-binding proteins, factor 1, MEF2 (myocyte enhancer factor 2), and Nf?b etc. (70,(72)(73)(74). It can also suppress small amounts of RNA involved in GLUT-4 expression such as miR-21a-5p, miR-222-3p, miR-29c-3p, and miR-133a-3p etc (75)(76)(77)(78). In addition, a variety of stress-inducing substances and products such as p38 MAPK, JNK / SAPK, PKC (protein kinase C), sorbitol, and hexosamine are all produced by oxidative damage and may suppress GLUT-4 (29). Therefore, reduced expression / activation of GLUT-4 is one of the major molecular mechanisms by which oxidative stress induces insulin resistance and contributes to the formation of DM (15).
7. c) Insulin Signaling Pathways
Insulin resistance with DM occurred due to any impairment in insulin signaling pathways (79,80). In appreciation of insulin sensitivity was introduced novel therapeutic variables of the insulin signal (80). Oxidative stress can disrupt normal IST (insulin signal transduction) at various levels including IR, IRS-1 and IRS-2; PI3K enzyme and Akt signature methods (81)(82)(83)(84)(85)(86). T2DM-induced oxidative stress and IST substances in
( D D D D )the brains of diabetic mice caused by Balbaa and colleagues in 2017 (87). They found that oxidative stress significantly reduced the expression of an IST substance such as p-IRS, p-AKT, and GSK-3? in brain tissue with normal insulin signaling (87). Oxidative stress induced IRS-1 and IRS-2 serine phosphorylation, leading to disrupted IST (81,82). Free radicals can induce serine phosphorylation of IRS-1 and suppress normal IST using JNK / SAPK signaling methods (85). Other types of serine / threonine kinases such as Akt (or PKB), GSK-3, AMPK, and mTOR are also very sensitive to oxidative stress and may interfere with insulin signaling (100)(101)(102). Oxidative stress can also lead to IST impairment by down-regulating proteins involved in normal IST (87). IST substances such as Akt, IRS, IRS-1, and GSK-3 are under the influence of low free radicals regulated by oxidative stress thus interfering with insulin sensitivity leading to insulin resistance and DM (87 ). Therefore, IST disruption is another important link between oxidative stress and insulin resistance (81)(82)(83)(84)(85)(86)(88)(89)(90).
8. d) Inflammatory Processes
The inflammatory response is one of the main molecular mechanisms involved in the pathophysiology of insulin resistance, DM, and related complications (53,91,92). A separate study suggests that chronic inflammation is involved in the pathophysiology of insulin resistance DM (92)(93)(94)(95)(96)(97)(98). This type of mechanism can also establish other pathophysiologic mechanisms of DM such as beta-cell dysfunction (53,95). Animal experimental and pathological studies show that IR and inflammation are directly linked during T2DM development (24,42) Inflammatory mediators play an important role in improving IR and T2DM by activating various inflammatory responses such as IL -?. IL-a is an effective pro-inflammatory mediator that plays an important role in controlling various inflammatory mediators such as cytokines, adipokines and chemicals. It causes inflammation by binding to interleukin-1 receptor type I (IL-1RI) and reduces the expression of insulin receptor substrate-1 (IRS-1) at the ERKdependent writing level and the post-transcriptional ERK level (43)). IL-1? production is largely controlled by dietary stress caused by diet. Other experimental studies have been performed on a variety of experimental animals to investigate the presence of various inflammatory responses in ?-cells that indicate that IL-? plays a key role in activating other inflammatory mediators such as cytokines and chemicals (21,22). in cell-cells of pancreatic islands due to impaired insulin secretion occurs in ?-cells of pancreatic islands. In this way IL-? also plays an important role in causing inflammation of the tissues of the body because it reduces the ability of the insulin receptors to respond to glucose which ultimately leads to the formation of IR in borderline tissues. IL-6 is another mediator that can be positively linked to IR [19 -22]. IL-6 not only inhibits the metabolism of non-oxidative glucose (120,121) but also suppresses the activity of lipoprotein lipase which increases plasma levels of triglycerides [23]. In addition, IL-6 also activates cytokine signaling proteins (SOCS) (101,108) that can inhibit cytokine transcriptional factor activation of the insulin receptor [26] causing IR development in borderline tissue.
TNF-? is another mediator in which TNF-? improved interactions between IR and T2DM (122,123) Experimental studies show that TNF-? expression increases in obese animals that modulate insulin action (135). From previous studies it has been found that serum levels of TNF-? are positively correlated with IR pathophysiology (135,136) indicating that TNF-? is also a key factor contributing to IR development. There are many monocytes, macrophage activity and mediators such as CX3CL1 (fractalkine), CRP 4 Oxidative Medicine and Cellular Longevity, IL-18, MCP-1 (monocyte chemo attractant protein-1), resistin, PAI-1 (plasminogen activator inhibitor -1), E-selectin, and IFN-? (interferongamma)-induced IR (91,(94)(95)(96)(97)(98).
Therefore, by making inflammatory processes a therapeutic approach for the management of diabetes (93,103,104). Several studies have reported the importance of anti-inflammatory agents in glucose homeostasis. For example, Goldfine and colleagues in 2010 examined the effects of sugar reduction salsalate (salicylate prodrug) and reported that it was effective in
( D D D D )reducing HbA1c and fasting plasma glucose in T2DM patients (105). Clinical trials have been performed with agents to reduce oxidative stress. Oxidative stress is a major inflammatory event as it stimulates the formation of monocytes and macrophages that promote inflammatory responses involved in insulin resistance and DM (103,106,107). It also regulates the expression of pro cytokines and thus enhances inflammatory mediators (88,108). Thus, free radical-induced inflammation is one of the possible links between oxidative stress and insulin resistance (99).
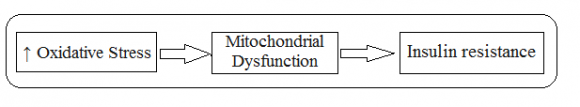
9. e) Mitochondrial Dysfunction
Mitochondria are cellular organelles that play a key role in energy production, reactive oxygen species (ROS), mediator signaling (130)(131)(132)(133), apoptosis (9,10) calcium storage, heat production, and cell survival and act as part of the signal signal pathways (109,110). Mitochondria are major sources of ROS (134) production that cause mitochondrial dysfunction, insulin resistance and DM (111). Oxidative stress is an important factor contributing to mitochondrial dysfunction (112), which impairs mitochondrial function by altering normal MRC activity, reducing mitochondrial respiratory capacity, increasing proton leakage to MRC, which alters potential fluid differentiation internal mitochondrial., and reduced the integrity of the mitochondrial layers (113)(114)(115). These processes can occur in pancreatic islands and / or systematically in adipocytes and muscle tissue (116). The normal process of glucose uptake depends largely on the body's function of healthy mitochondria that produce the energy needed to receive glucose from borderline tissues (117). Thus, mitochondrial dysfunction significantly reduces ATP cell production and interferes with cellular glucose uptake (88). As a result of these cells they fail to take up circulating glucose in response to insulin leading to insulin resistance (88,16,118). In addition, oxidative stress can disrupt normal mitochondrial function by increasing the production of mitochondrial fatty acid oxidation and DAG (diacyl glycerol), which also stimulates many serine / threonine kinases leading to IST impairment (88). Thus, stressdependent mitochondrial dysfunction is another molecular mechanism by which free radicals induce insulin resistance (88,116,118). Oxidative stress and mitochondrial dysfunction have two interactions where both can produce energy (119).
10. Conclusion
Diabetes is a metabolic disorder due to hyperglycaemia that can be completely cured but not normally controlled. There are many factors that contribute to the increase and progression of diabetes. Factors can be genetics, stress, obesity, and unhealthy lifestyle etc. But above all oxidative stress plays an important role in the development and progression of diabetes. Oxidation is a chemical process within the human body that leads to the production of free radicals. These oxidative reactions produce free radicals that slowly damage cells and organs by removing inflammatory mediators (such as TNF-?, cytokines, adipokines and chemicals etc.) and causing damage to cell organelles such as mitochondrial damage, damage of ribosomal, nucleus damage etc. leading to insulin resistance. Among other things, oxidative stress increases the rate of disease progression by interfering with insulin signaling pathways resulting in a decrease in insulin sensitivity. Oxidative stress increases apoptosis necrosis leading to beta cell dysfunction leading to insulin secretion. Antioxidants play a very important role in eliminating free radicals. They bind with free oxidative radicals and remove them from the body by making it harder. Combining antioxidant treatment with standard hypoglycaemic medications will increase recovery rate and antioxidant therapy will help address diabetes problems such as nephrotoxicity, neuropathy and retinopathy, which may be caused by oxidative stress.