1. Isolation and Partial Characterization of Virulent
Phage Specific against Pseudomonas Aeruginosa Zahra Zahid Piracha ? , Umar Saeed ? , Aqsa Khurshid ? & Waqas Nasir Chaudhary ? Pseudomonas aeruginosa is an opportunistic pathogen, frequently associated with nosocomial infections worldwide. Multiple drug resistance has been reported in previous studies against pathogenic P. aeruginosa and the biofilm which makes use of antibiotics futile. Bacteriophages specific for P. aeruginosa can prove to be a new therapeutic approach for controlling infections and biolfilm contamination against this pathogen. The aim of our study was to isolate and partially characterize virulent phage specific for P. aeruginosa from sewage water. Different parameters of which make phages as suitable candidate towards future therapeutics were also investigated. Phages having lytic life cycle, high burst size and thermally more stable proposed themselves as effective therapeutic candidates. In this study, a virulent phage was isolated from sewage water having burst size of 1036 and latent period of 21 minutes. This phage has narrow host range and shows the remarkable thermal resistance and is viable up to 60 ºC. Sincere efforts in term of identification, isolation, purification and characterization of multiple types of phages Introduction seudomonas aeruginosa is Gram negative, rod shaped opportunistic pathogen of animals, ubiquitous in nature [1]. According to United States National Nosocomial Infection Surveillance System, it accounts for 16.1% of all nosocomial infections and ranked second among Gram negative pathogens [2]. Healthy adults rarely encounter its infection but main target is people having compromised immune system including HIV infections. The infection ranges from self-limiting folliculitis to life threatening bacteremia, wound related morbidity, septicemia, skin infections, otitis media, ecthymegangrenosum, the black necrotic lesion, endocarditis, corneal ulceration and device related infections [3][4]. It is the third leading cause of 12 % of hospital-acquired urinary tract infections, upper and lower respiratory tract infections like cystic fibrosis that is associated with high mortality rate in immune-compromised patients [4]. Gender-wise prevalence showed 61.78% male and 38.22% females were infected by P. aeruginosa [3]. Souli and colleagues (2008) [5] published data from 23 countries on the European Antimicrobial Resistance Surveillance System and it was shown that 18% of all isolates were multidrug resistant P. aeruginosa strains. Aman Ullah and colleagues (2012) [6] carried out a study in Islamabad and showed that P. aeruginosa is 94% resistant to Chloramphenicol, 88% to Colistin /sulphate, 73% to Tetracycline and 3% to Imipenem. The resistance against the newly tested drugs is still evolving as P. aeruginosa is highly resistant to antibiotics, both at the genetic level and as a result of living in multilayered and complex biofilm [7].
Bacteriophages are bacterial viruses that cause lethal effect on bacteria. They have genetic material in the form of either DNA or RNA (single or doublestranded), encapsidated by a protein coat [8]. Therapeutic role of the phages was interrupted by the advent of antibiotics however the emergence of multidrug resistant bacteria and adverse effects of antibiotics led to renewed interest in phages as therapeutic agent. [9]. Phages have several advantages over antibiotics and other antimicrobial agents, such as host specificity, no side effects, and multiplication in the presence of their hosts [8]. Phages are highly specific and as they are living entity so they evolve with the evolving bacteria [10]. The specificity of phages sometimes may be considered as a possible disadvantage because there are much more pathogenic bacteria than expected to be targeted. To address this problem, a cocktail of phages should be prepared [8]. The Food and Drug Administration of the United States of America recently approved some phages as safe for food products to control Listeria infections [11]. Animal tests of phage therapy are being conducted for treatments of various bacterial infections, and many lytic phages have been isolated and tested for such applications. Phages have been used to treat the Escherichia coli infections and P. aeruginosa infections in mouse models and in guinea pigs and efficient results have been obtained [12].
Approximately 10^30 bacteriophages are present in the environment but only about 300 phages have been characterized [13][14]. In the pre antibiotic era, phage therapy was failed because uncharacterized phages were used so in order to use them fully, it is important to isolate and characterize new phages especially in light of the observation that most of the disease-causing organisms live in matrix-enclosed environments called biofilms that inherently show increased resistance toward all antibiotics [15][16]. In the present study, we have reported the isolation and partial characterization of a virulent phage specific for P. aeruginosa from sewage water in Islamabad and evaluated the different parameters of phage that makes it suitable candidate for future therapeutics.
2. II.
3. Materials and Methods
4. a) Identification of bacterial isolate
Pure cultures of bacterial strain were obtained from Microbiology laboratory of Kahuta research lab hospital, Islamabad. The bacteria were already resistant to Chloramphenicol, Colistin, Cotrimoxazole, Tetracycline and Aztreonam. After overnight incubation of bacterial strain, microbiological methods such as colony morphology and Gram staining were used for identification of bacterial strain [17].
5. b) Phage enrichment and isolation
Sewage water was taken from Sewage treatment plant I-9 Islamabad. Sewage water was centrifuged at 10,000 rpm for 10 min to remove algal cell and sewage debris. To enrich the phage population, above prepared sample concentrates (5 ml) were added to a 30 ml log phase P. aeruginosa. Enriched cultures were incubated overnight at 37 °C with shaking at 150 rpm. Chloroform (1%) was added to 1.5 ml of sample to disrupt bacterial cell and release phages and then centrifuged at 14,000 rpm for 10 min at 4 °C. The supernatant was filtered using 0.45 and 0.20 mm (Minisart, SalotriusStedim Biotech) syringe filters and transferred to a new tube. Phage isolation and detection was carried out by plaque assay on LB agar plate (tryptone 10 g/L, yeast extract 5 g/L, NaCl 10 g/L, agar 15 g/L) with soft agar (tryptone 10 g/L, yeast extract 5 g/L, NaCl 10 g/L, agar 0.7%) . Overnight bacterial culture and phage sample were mixed in 0.7% LB soft agar (tryptone 10 g/L, yeast extract 5 g/L, NaCl 10 g/L, agar 0.7%; at a temperature of 45 °C) and poured over LB plates. On solidification plates were incubated overnight at 37 °C and examined for plaques the following day. For a negative control, phage alone was added to the molten agar. Well isolated plaques were serially propagated until a single phage type was obtained. The purified phages were then stored in SM buffer (100 mMNaCl, 8 mM MgSO4, 50 mM, Tris-HCl [pH 7.5]), and 0.002% w/v gelatin at 4 °C with the addition of 7% dimethyl suloxide (DMSO) at -80 °C. [18] c) One step growth experiment One step growth curve experiment was performed to determine latent period and burst size of phage [19][20]. In brief, 50 ml of P. aeruginosa culture was incubated to mid exponential phase having O.D 600 0.6 and cells were harvester via centrifugation. The pellet was re-suspended in 0.5 ml of LB media and mixed with 0.5 ml of the phage solution having plaque forming unit (pfu) of 2.75x10^9. This mixture was allowed to stand for 3 minutes at 37 °C so that phages adsorbed to the host cells. Mixture was then centrifuged at 13,000 rpm for 2 minutes to remove the free phage particles. The pellet was re-suspended in 100 ml of LB medium and culture was incubated at 37 °C with shaking. Samples were taken after every 3 minutes for 45 minutes and after centrifugation at 13,000 rpm for 1 minute, subjected to determination of phage titer via plaque assay.
6. d) Analysis of calcium ion effect on phage adsorption
In order to measure the effect of divalent metal ions on phage adsorption rate CaCl 2 was used [21]. The 50 ml of P. aeruginosa overnight culture having O.D 600 0.6 was divided in two autoclaved flask 25 ml each. One flask was inoculated with phage 500 ?l having pfu of 2.75x10^9 (control), while the second flask was inoculated both with 500 ?l phage (pfu 2.75x10^9) and 250 ?l of 1 M CaCl s and incubated with constant shaking at 90 rpm at 37 °C. Samples were taken from both flasks at different time intervals of 0, 10, 20, and 30 minutes. Samples were centrifuged at 13,000 rpm for 3 minutes to sediment the phage adsorbed bacteria. The supernatant was assayed for unabsorbed phages via double layer plaque assay method and counts were compared with the titer of control. e) Thermal stability analysis Thermal stability tests were performed according to the method described by Suarez [21]. Phage filtrates were taken in microcentrifuged tubes and treated under different temperatures at 37 °C (control), 40 °C, 50 °C, 60 °C and 70 °C for 20 minutes, 40 minutes and for 1 hour. After incubation plaque assay was performed for each treated sample. Results were compared with control. P. aeruginosa culture 1 ml was inoculated in 2 flasks having 150 ml LB media. The phage supernatant 500 ?l was introduced in one flask and other flask was treated as control and both the flasks were incubated in shaking incubator at 37 °C. The O.D 600 readings were taken after every 2 hours for 24 hours using spectrophotometer. The values were compared with control.
7. g) Host range
The host range of the isolated phage was checked on the range of clinical pathogenic bacterial L, latent phase; R, rise phase; P, plateau phase.
8. d) Calcium Ion Effect on the Adsorption Rate of Phage
Divalent ions often affect the adsorption of phages. In order to check this, CaCl 2 1mM was added in P. aeruginosa phage mixture and number of free phages left in solution were detected after 10 minutes interval for 30 minutes by using plaque assay. Data analysis showed that significant differences existed between the control and calcium ion treated group. Results obtained show that calcium ions stabilize the process of adsorption and infectivity is increased due to addition of CaCl 2 . Infection of bacteria with the phages was monitored for 24 hours. The bacterial reduction caused by phages was compared with control. Phage infection produced a drastic decrease of the P. aeruginosa culture as compared to control as it is evident from the graph. However, a constant increase in OD 600 was seen after 16 hours this is most probably due to growth resistant phages. The susceptibility of the phage was investigated with clinical strain of P. aeruginosa isolated from various sources and results show that the phage hasnarrow host range.
9. Discussion
P. aeruginosa is a nosocomial pathogen and leading cause of health care associated infections. It is an emerging multidrug resistant pathogen around the globe and also in Asia [2]. Way of acquiring resistance specifically the chromosome encoded efflux and low outer membrane permeability accounts for its high resistance and makes use of antibiotics futile. The biofilm induces the resistance and restricts the diffusion of the antibiotic which makes most of the antimicrobial agents less effective. It can also acquire additional resistance genes from other organisms via plasmids, transposons and bacteriophages [7]. Imipenem was considered one of the effective antibiotics however Lautenbech and colleagues in 2010 detected the imipenem resistant P. aeruginosa strains. All these evidence showed an alarming situation and demands an alternative treatment for P. aeruginosa infections [6, 23].
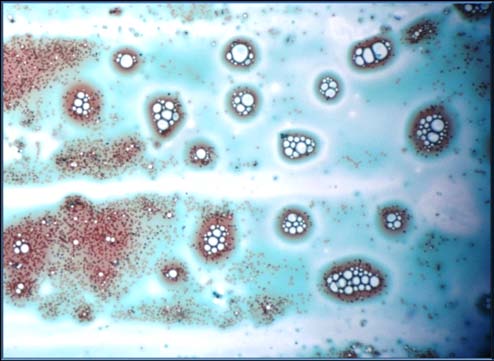
In this study, our results showed that a newly isolated bacteriophage can lyse actively growing P. aeruginosa. Phages are found with the natural habitat of their host cells such as sea water, sewage water/ sludge ponds etc [1]. They are host specific and evolve along with their host [10]. Phage was isolated from sewage water. Sewage in general, contains a large diversity of microbes due to contamination from fecal and hospital drainage water. This newly isolated phage was highly lytic and produced large plaque of 4 mm. Phage establishes an infection in the host when it gains entry into the host after interaction with the bacterial receptor [24]. Many phages have been found to be greatly specific for their receptors present on the host cell surface. They show no interaction with receptors having a different structure. The specificity turns out to be the basis of phage typing methods used for the identification of bacterial strains. The results obtained clearly showed that our virulent phage for P. aeruginosa was highly specific. Results indicated that the phage may be using a common receptor.
One step growth curve explains all the stages involved in multiplication of phages. From this curve, latent period and burst size are determined. Latent period or incubation period is the time which the phage requires inside the bacterial cell and is 21 minutes in case of our isolated phage. The optimum latent period leads to high phage fitness. Rise phase is the time when infected cell begin to lyse and plateau phase is time when the infectious centers is due to only single virion [19]. Phage burst size is average number of phage bacterial cells produced per bacterial cells upon infection. The burst size of phage is 1036 particles per bacterial cell. Wang [25] described that the duration of the latent phase correlates with the burst size. The longer is the latent phage the bigger is the burst size of the phage. The number of phages liberated upon infection is quite high as compared to early reported phages.
The factors which influence the infectivity of phage in-vitro are temperature and divalent cations. The physiological state of host also plays an important role in reproduction of phage. The infection of the virion starts when it binds to host bacterial cell. In phage host interaction, calcium might be playing an important role in infection cycle [19] .Calcium ion stabilizes the weak interaction of virion with receptors or they may change the structure of the receptor to make the accessibility of phages smoothly. Ions have an electrostatic effect in the interaction of phage bacterium systems [26]. Different amount of calcium gives different infectivity.
The physical parameters like the temperature not only accelerate the adsorption of phages but also have direct effect on metabolic activities of the host [26]. Extremely thermal resistant phages have been isolated from thermal habitats and also in other aquatic environments [27]. Our results implied that phage we have isolated is active on high temperatures. The phage remained active till four months at 4 °C and it remains viable up to 60 °C after 1 hour incubation. The maximum infectivity was observed at 37 °C and least infectivity was observed at 60 °C.
The phage we isolated also causes decrease in turbidity of P. aeruginosa culture and this behavior was obvious for 16 hours as compared to control. There was maximum bacterial destruction during this time period but some bacterial cells show resistance to virus infection. This may be because of host bacterium adaptation as bacterial replication was going on meanwhile and bacterial resistant cells survived and though they were less in number but they started multiplying after about 16 hours and it was time when turbidity of culture increases as shown in figure 6. This
Volume XIV Issue I Version I Year ( )
10. 2014
type of behavior can be a hindrance in phage therapy. Some studies shown that phage resistant bacteria losses their virulence factor because those virulence can be a site for phage infections. Such loss of virulence factor in a phage resistant bacteria mutant has been demonstrated in fish pathogens [28]. Due to resistance, the fitness of bacteria reduces and it renders it competing unfavorably with its phage sensitive ancestors. The gradual increase in optical density can also be attributed to some other factors like bacterial debris and this might be a hindrance in exposition of actual results.
V.
11. Conclusion
Viral diseases are increasing day by day in world and it is anticipated that soon hepatitis viruses would emerge as most dangerous viral pathogens [29,30]. But on the other hand nature has selected many viruses to kill various other pathogens. . Nature has many hidden remedies against multiple disorders such as cardiovascular diseases, metabolic disorders, cancer, chronic inflammation and many others); there is strong need to discover therapeutic potentials of natural items. Many studies showed elevated activation of various cellular proteins cause cancer proliferation, which can be further inhibited by potential inhibitors (31-Partial characterization of the virulent phage showed that it is highly efficient in lysing Pseudomonas aeroginosa, as it has shown some outstanding aspects including rapid growth nature, high thermal stability and optimum latent period. All these characteristics make this phage very promising for possible application in eradication of Pseudomonas aeroginosa contaminations and treatment of Pseudomonas aeroginosa infections. Phage has shown narrow host range, so for the broad-spectrum elimination of bacteria; a "cocktail" with a pool of lytic phages might be more useful against present and other bacterial strains. A better understanding of phages and lytic enzyme biology could facilitate development of novel future therapeutics against multiple drug resistant bacteria.
Volume XIV Issue I Version I
![Isolation and Partial Characterization of Virulent Phage Specific Against Pseudomonas Aeruginosa f) Bacterial reduction assay strains of P. aeruginosa isolated from blood, wound, urinary tract, ear and pus, Streptococcus pneumoniae, Escherichia coli and Klebsiella pneumoniae. All the tested bacterial strains were clinical pathogens obtained from Microbiology laboratory of Kahuta research lab hospital, Islamabad. Susceptibility of the phage was tested via the spot on lawn technique[22]. Plates were incubated upside down overnight and checked for any plaque against the negative control i.e., uninfected lawn.III. Resultsa) Identification of bacterial isolateIn this study, P. aeruginosa was identified via Gram staining and colony morphology. Gram staining showed that it is Gram negative bacillus.](https://medicalresearchjournal.org/index.php/GJMR/article/download/521/version/100235/1-Isolation-and-Partial-Characterization_html/3909/image-2.png)
| Sr. | Color Margin Shape Elevation Luminous | Texture Size |
| No | behavior |
| 100.00% | |||||||||
| of surviving PFU (%) Percentage | 0.00% 20.00% 40.00% 60.00% 80.00% | 37 Degrees 40 Degrees 50 Degrees | |||||||
| 10 | 20 | 30 | 40 | 50 | 60 | ||||
| Incubation time in minutes | |||||||||
| 5 | |||||||||
| Bacterial O.D600 | 0.000 0.100 0.200 0.300 0.400 0.500 0.600 0.700 | 0 | 5 Control Flask Having Phage 10 | 15 | 20 | 25 | 30 | Medical Research Volume XIV Issue I Version I ( D D D D ) | |
| Time (Hours) | Global Journal of | ||||||||
| Sr. No Source of bacteria | Clinical strain | Host range (+,-) | |||||||
| 1. | Pus sample | P. aeruginosa | + | ||||||
| 2. | Blood sample | P. aeruginosa | - | ||||||
| 3. | Urinary tract sample | P. aeruginosa | _ | ||||||