1. Introduction
n the basis of clinical and histopathological findings, Pindborg defined Oral Submucous Fibrosis as "an insidious chronic disease affecting any part of the oral cavity and sometimes the pharynx. Although occasionally preceded by and or associated with vesicle formation, it is always associated with a juxta epithelial inflammatory reaction followed by a fibro elastic change of the lamina propria with epithelial atrophy leading to stiffness of the oral mucosa and causing trismus and inability to eat".
It is a slow, progressive fibrotic disease causing fibroelastic change and inflammation in the oral mucosa, leading to inability to open the mouth, swallow or speak 1,2 . This was accredited to the accumulation of inelastic fibrous tissue in the juxta epithelial region of the oral mucosa, along with concomitant muscle degeneration 3 . The most common site to be involved was found to be the buccal mucosa, although other parts of the oral cavity were also found to involve, including the pharynx 4 .
These reactions may be the result of direct stimulation from exogenous antigens like Areca alkaloids or changes in the tissue antigenicity that may lead to an autoimmune response.
2. II.
3. Epidemiology
Most common prevalence was found among the Indians, ranging from 0.2% to 1.2%. A survey revealed an overall prevalence of up to 4% in kerala 5 . Amongst the reported cases 0.5% was found to be in women 6 . The reason for the rapid increase of the disease is reported to be due to an upsurge in the popularity of commercially available areca nut in south asia 7 .
III.
4. Etiology and Pathophysiology
The etiology of OSMF is still not fully unwinded. It is considered to be a multifactorial disease. According to Liao, the areca nut in betel quid plays a major role in the pathogenesis of OSMF 8 . There was no significant evidence to relate the habits of smoking or alcohol consumption alone in the pathogenesis of the disease 9,10 . Arecoline, an active alkaloid found in betel quid, stimulates fibroblasts to increase production of collagen by 150% 11 . Chung-Hung in 2006, studied that arecoline was found to elevate mRNA and protein expression of cystatin C, a non glycosylated basic protein consistently upregulated fibrotic diseases, in a dose dependent manner in persons with OSMF 12 .
Yet another hypothesis grips on the fact that, the high copper content of areca nut acts as an initiating factor in OSMF. This was suggested by the fact that, soluble copper levels in oral fluids significantly increases after chewing areca nut for 5-30 minutes 13 .
Other factors thought to cause OSMF include iron and vitamin B complex deficiency that derange the repair of inflamed oral mucosa, leading to defective healing and resultant scarring. As a result, the oral mucosa becomes more prone to the effects of areca nut and chilies.
The role of chilies and spices in the list of etiological agents is still a topic of debate among the scientific community. It was shown that the capsaicin in chilies stimulates the widespread palatal fibrosis in rats 14 . However, the incidence of OSMF was lower in Genetics is believed to play a role in OSMF patients, which leads to immune system changes. People without the betel nut chewing and chilly ingestion were reported to have OSMF 16 . Mutations in APC gene and low expression of wild type TP 53 tumor suppressor gene in affected patients, increased the risk of malignant transformation 17 .The increase in CD4 cells with HLA-DR in OSMF tissues suggest that lymphocytes are activated and number of langerhans cells increased. The presence of these immunocompetent cells and with increased CD4 to CD8 ratio in OSMF tissue, suggest an ongoing cellular immune response resulting in imbalance of immune regulation and an alteration in local tissue architecture. These reactions can be from the direct stimulation from exogenous antigens or of changes in tissue antigenicity that leads to an autoimmune response 18 . Increased levels of proinflammatory cytokines and reduced antifibrotic interferon gamma (IFN-gamma) in patients with OSMF were demonstrated 19 .
IV.
5. Clinical Features
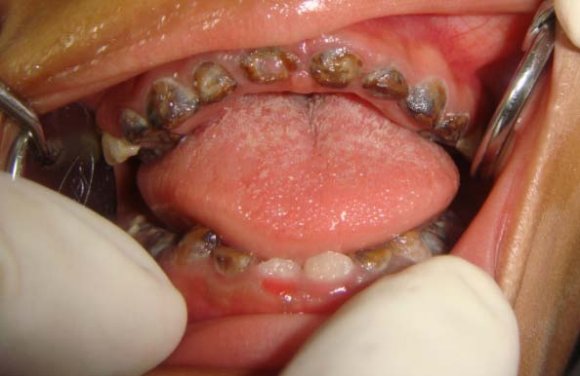
OSMF was found to be predominant in females 16 . The mean age was 43 years. Burning sensation and discomfort in the oral mucosa during mastication was the most common complaint amongst the reported cases. There is associated depapillation of the tongue (Fig: 1). Progressive changes including difficulty in mastication, reduced salivation, dysphasia, pain in the ears and loss of auditory acuity due to stenosis of the pharyngeal end of Eustachian tubes.
In advanced cases, the jaws become inseparable and totally inelastic and plastic and nutrition can be maintained only by pushing the food into mouth. The buccal mucosa is frequently ulcerated and secondarily infected consequent to ischemia and constant pressure of the mucosa against the buccal aspect of the teeth.
Pindborg provided staging criteria for OSMF 20 . He divided the stages according to the clinical presentation of the disease as: Early lesions demonstrate blanching of oral mucosa and older lesions presents with vertical and circular palpable fibrous bands in the buccal mucosa and around the mouth opening or lips, resulting in mottled, marble like appearance(Fig: 4) of the mucosa.
This stage is characterized by reduced mouth opening, stiff and small tongue, fibrotic and depigmented gingiva, shrunken bud like uvula.
Stage 3: speech and hearing deficits may occur as a part of the sequel. Histological examination reveals severely atrophic epithelium with complete loss of rete ridges. Varying degrees of epithelial atypia may be present. The underlying lamina propria exhibits severe hyalinization, with homogenization of collagen. Cellular elements and blood vessels are greatly reduced 18 .
V.
6. Investigations
Clinical presentation of the disease plays an upper hand in the diagnosis phase. Other investigations include complete hemogram, toludine blue test, incisional biopsy and immune fluorescence tests 21 .
7. VI.
8. Management
The earlier the treatment begins, better it is for the patient. The treatment modes depend upon the state of the disease at the time of presentation. If it is detected at a very early stage, cessation of the habit would provide sufficient relief. But moderate to severe stages of OSMF are almost always irreversible. Usually the disease is very resistant to treatment. The proposed treatment regimens aims hinder the progression of the disease process. Submucosal injected steroids and hyaluronidase, placental extracts, oral iron preparations and topical vitamin A and steroids are some the agents that have been used 22 . All of these therapies are usually palliative. Surgical treatment includes simple excision of fibrotic bands, split thickness skin grafting following bilateral temporalis myotomy or coronoidectomy 11 . The use of oral stent as an adjunct to surgery to prevent relapse of the fibrotic bands has also been studied 23 . Other treatment modalities include administration of Antoxid OD for 6-8 weeks, Lycored OD for 6-8 weeks and physiotherapy for improving mouth opening 22 .
9. VII.
10. Malignant Transformation
OSMF is a well recognized potentially malignant disorder of the oral mucosa. Simultaneous occurrence of oral leukoplakia and OSMF is demonstrated to carry a IX. higher risk for malignancy than with OSMF alone which amounts from 4-13% 24 .
11. Volume
VIII.
12. Conclusion
The incidence of OSMF is on the rise with the popularity of commercially available betel nut products. It also carries a significant morbidity rate from oral cancer. So it is desirable that OSMF is diagnosed as early as possible. At best, it is palliate the symptoms of OSMF. In palliative care, the patient is the focus of treatment, not the disease. A stepwise approach to OSMF management is advocated, with the level of entry into the treatment algorithm being dictated by the disease severity and response to treatment. Although clinicians strive to achieve lesion resolution and primary outcomes of therapies aims to concentrate on symptom reduction and improvement of quality of life. Intervention studies and public health awareness programme linked with OSMF condition and habits may prove the best way to control disease process at the root level.